Beyond Biosimilarity: The 2026 Strategic Blueprint for Day 1 Interchangeability
Executive Summary
For over a decade, the Interchangeable designation was the most expensive badge of honor in the biosimilar industry, typically requiring a multi-million dollar clinical switching trial. As of early 2026, the regulatory wall has collapsed. Following the FDA’s pivotal June 2024 guidance and the comprehensive Biosimilar Reform Initiative of October 2025, the industry has shifted from a clinical-volume requirement to a Scientific Justification standard. This article explores how modern Clinical Pharmacology—specifically Population PK (PopPK) and Virtual Switching simulations—now serves as the primary evidence for interchangeability.
1. The 2026 Strategic Landscape: Interchangeability as a Day 1 Requirement
In the current market, Biosimilarity is the baseline, but Interchangeability is the commercial weapon. As PBM (Pharmacy Benefit Manager) negotiations become more aggressive, the ability for a pharmacist to automatically substitute a reference product (RP) with a biosimilar is the difference between market dominance and a stalled launch.
Today, successful developers architect their programs to secure the interchangeable label on Day 1 of approval, bypassing the clinical switching trial in favor of a robust Clinical Pharmacology and Analytical package [1, 2].
2. The Historical Burden: Anatomy of the Switching Trial
Under the original 2019 framework, interchangeability required proof that switching back and forth would not present greater risks in terms of safety or diminished efficacy [2].
The 3-Switch Protocol
The gold standard stress test was the randomized multiple-switch crossover study. The theory was that alternating exposures might prime the immune system to recognize subtle molecular differences, leading to neutralizing Anti-Drug Antibodies (ADAs).
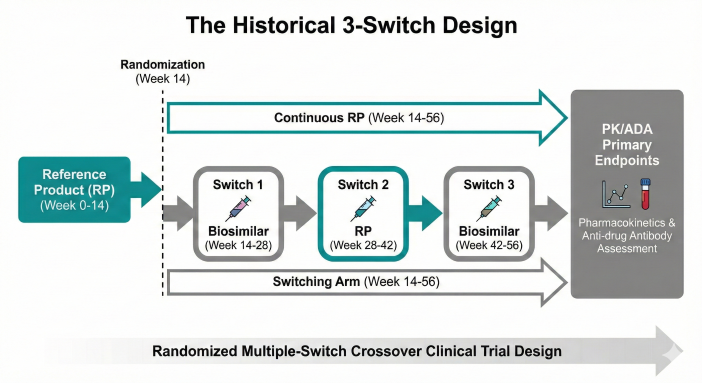 Figure 1: The Historical 3-Switch Design. A randomized crossover architecture formerly used to evaluate immunogenicity stability via repeated product transitions.
Figure 1: The Historical 3-Switch Design. A randomized crossover architecture formerly used to evaluate immunogenicity stability via repeated product transitions.
A typical design included:
- Lead-in Phase: Patients were stabilized on the Reference Product for 12–16 weeks.
- The Switching Arm: Patients underwent a minimum of three switches (e.g., RP → Biosimilar → RP → Biosimilar).
- The Non-Switching Arm: Patients remained on the Reference Product as a control.
Case Study: The Complexity of VOLTAIRE-X (Adalimumab-adbm)
The VOLTAIRE-X trial (NCT03210259), which supported the interchangeability of Cyltezo, serves as the definitive case study for this clinical-heavy era [3].
The study required 238 patients to be observed for 52 weeks. While successful, the trial required nearly a year of clinical follow-up to confirm what analytical fingerprinting already indicated. Similarly, the AVT02 (Simlandi) program required a switching study of 550 patients to secure its interchangeable status [4]. In 2026, these studies are viewed as Scientific Relics—monuments to a time when clinical data was used as a costly substitute for analytical confidence.
3. The Regulatory Engine: PopPK as the Extrapolation Tool
Population PK (PopPK) is the specific regulatory engine that makes a switching waiver possible. It provides the mathematical bridge required to move evidence from one population or indication to another.
Historical Precedent: Infliximab-dyyb and Filgrastim-sndz
The FDA used the first biosimilars, such as Infliximab and Filgrastim, as case studies to support the extrapolation of data across indications [5].
The Evidence: Regulators used PopPK modeling to show that the clearance and volume of distribution of the biosimilar were identical to the reference product across different disease states. This pharmacometric bridging proved that the drug would behave the same way in a Crohn’s patient as it did in the RA patient studied in the clinic.
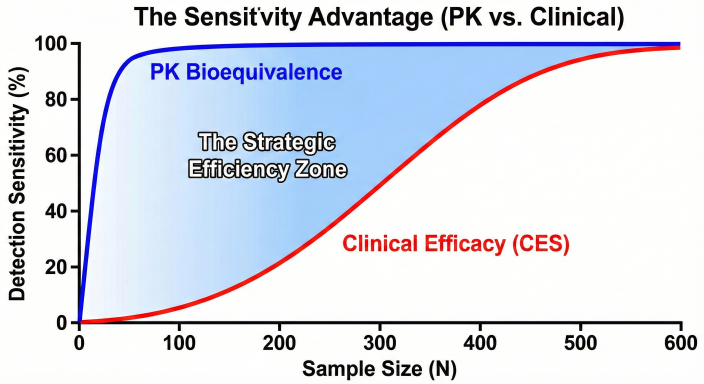 Figure 2: The Sensitivity Advantage. PK data offers significantly higher resolution for detecting product differences than coarse clinical efficacy endpoints.
Figure 2: The Sensitivity Advantage. PK data offers significantly higher resolution for detecting product differences than coarse clinical efficacy endpoints.
4. The “Virtual Switching” Whitepaper: Power of Evidence
While Virtual Switching is an industry term, its regulatory weight is grounded in the Model-Informed Drug Development (MIDD) pilot program. Organizations like the AAPS have advocated for PopPK simulation to replace physical switching studies [6].
The Logic: Instead of switching 200 real patients, developers use a validated PopPK model to virtually switch 10,000 diverse patient profiles. This provides a Power of Evidence that a small clinical trial could never reach, statistically proving that exposure remains within the bioequivalence (BE) margins of 80%–125% during a switch.
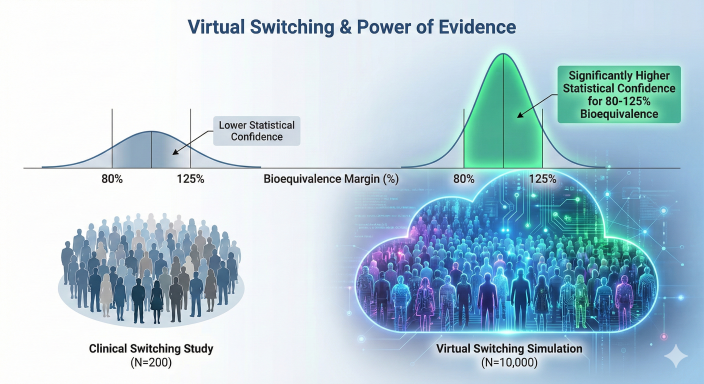 Figure 3: Virtual Switching vs. Clinical Observations. Visualizing the massive increase in statistical confidence provided by 10,000 simulated patient switches.
Figure 3: Virtual Switching vs. Clinical Observations. Visualizing the massive increase in statistical confidence provided by 10,000 simulated patient switches.
5. Strategic Blueprint for Module 2.7.2
To secure Day 1 Interchangeability, the Clinical Pharmacology summary must be architected as a Decision Narrative. The burden of proof has shifted from the clinic to the dossier.
| Component | Tactical Execution in the Dossier | Grounding & Precedent |
|---|---|---|
| Assay Resolution | Demonstrate that ADA assays are drug-tolerant and capable of detecting low-affinity antibodies. | AAPS Whitepaper: Critical for resolving fears of silent immune responses [6]. |
| Scientific Extrapolation | Detail the Mechanism of Action (MOA) identity across all licensed indications. | FDA Extrapolation Framework: Proven by Infliximab and Filgrastim case studies [5]. |
| PopPK Simulation | Execute Virtual Switching to prove bioequivalence across 10,000 simulated patients. | Oct 2025 FDA Reform: The technical engine replacing Switching Studies [2]. |
| MOA Identity | Prove identical glycosylation and oxidation profiles that drive immunogenicity. | Fingerprint Similarity: The analytical anchor of the 2024 Update [1]. |
Conclusion: The ClinPharm Advantage
In 2026, the biosimilar industry is no longer a race of clinical volume, but a race of strategic narrative. At ClinPharm Dev Solutions, we help our clients move from clinical-heavy to science-heavy strategies, ensuring that the interchangeable label is secured on Day 1.
Secure Your Day 1 Interchangeability
Is your 2026 program still planning a redundant $20M switching trial? Let’s architect a scientific waiver that saves capital and accelerates your launch.
Book a Strategic ConsultationAbbreviations
- ADA: Anti-Drug Antibody
- AUC: Area Under the Curve
- BE: Bioequivalence
- CES: Comparative Efficacy Study
- MIDD: Model-Informed Drug Development
- MOA: Mechanism of Action
- PopPK: Population Pharmacokinetics
- RP: Reference Product
References
- FDA Draft Guidance (June 2024). Considerations in Demonstrating Interchangeability with a Reference Product: Update.
- FDA News Release (Oct 29, 2025). FDA Moves to Accelerate Biosimilar Development and Lower Drug Costs (Biosimilar Reform Initiative).
- Schreiber, S. et al. (2022). VOLTAIRE-X: A randomized, double-blind, interchangeability study of adalimumab biosimilar Cyltezo.
- Feldman, S. R. et al. (2023). Assessing the Interchangeability of AVT02 and Humira: Results from a Multicenter Randomized Study.
- FDA Whitepaper (2025 Update). Application of the FDA Biosimilar Extrapolation Framework: Case Studies in Infliximab and Filgrastim.
- AAPS Whitepaper (2014/2017). Systematic Verification of Bioanalytical Similarity Between a Biosimilar and a Reference Biotherapeutic.