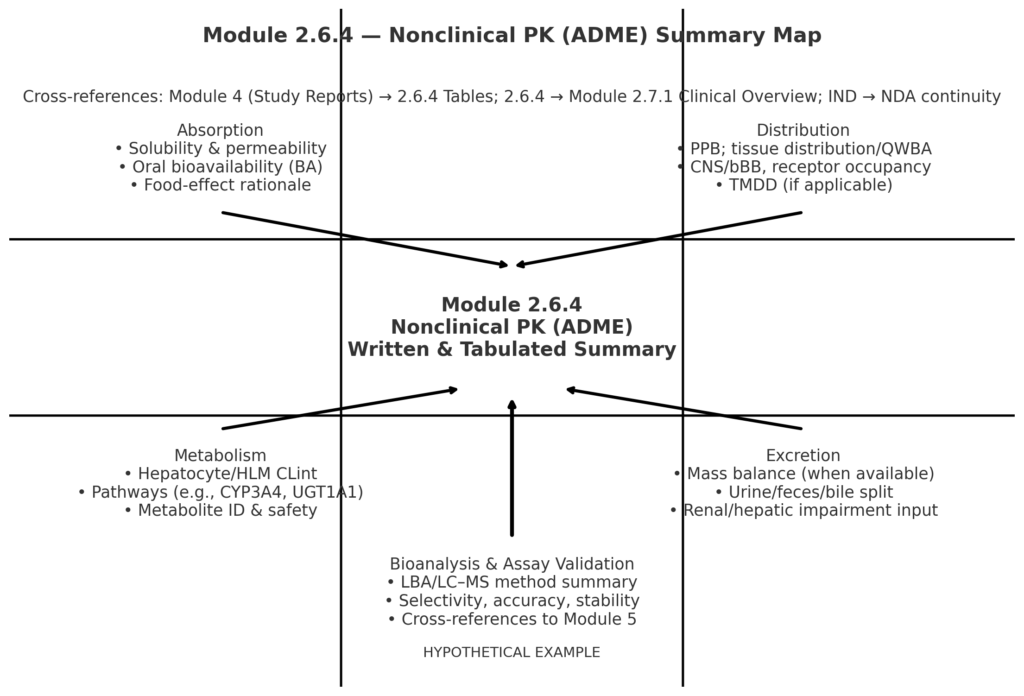
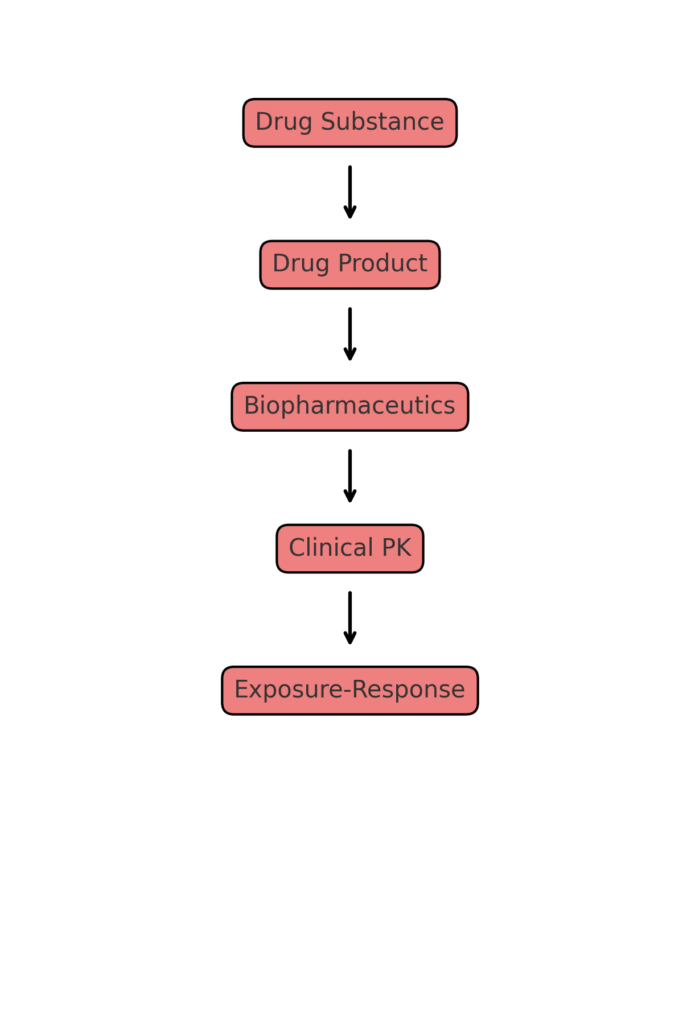
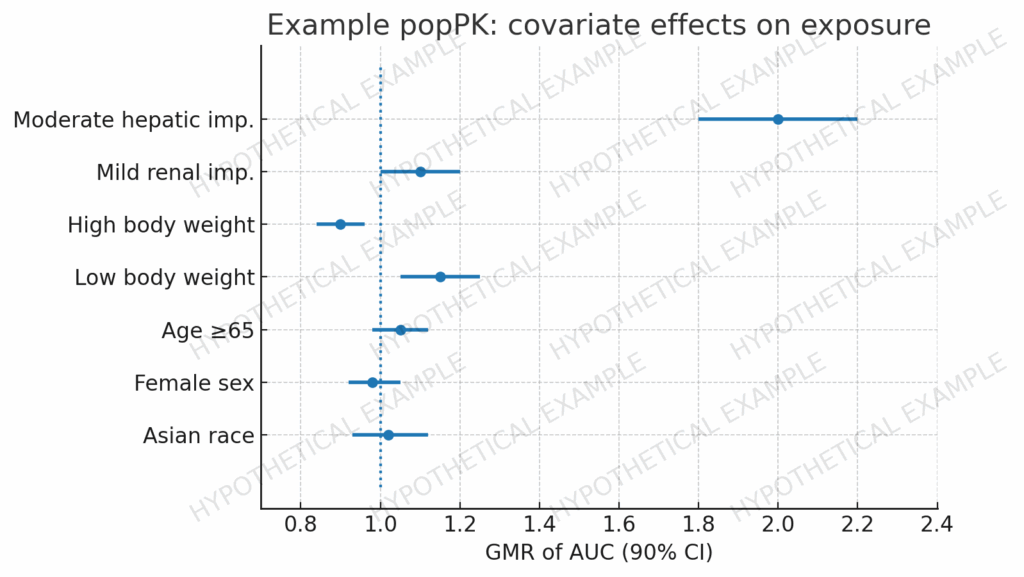
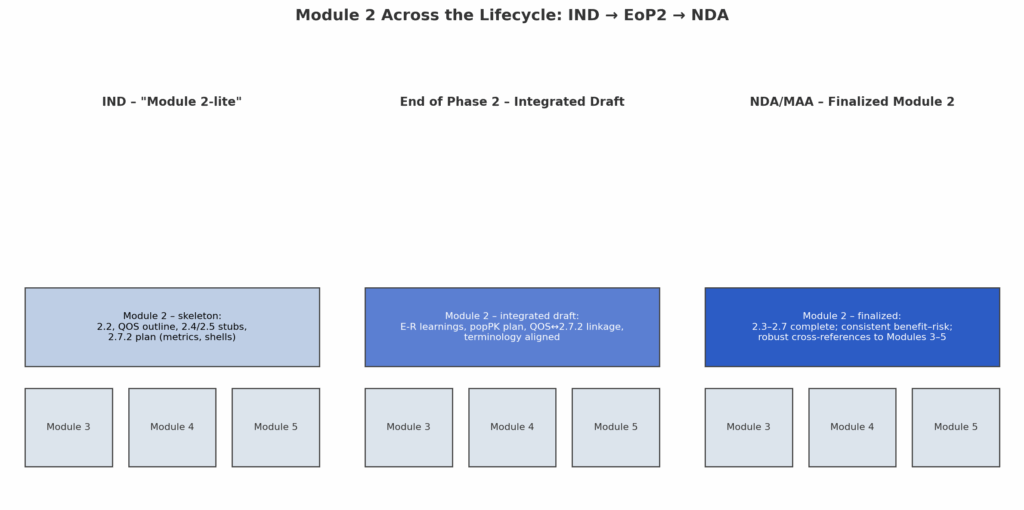
Modeling & Simulation as Regulatory Evidence in the CTD: Practical Case Studies Across PopPK/PD, PBPK, E–R, and QSP
Model-informed analyses are increasingly recognized by regulators and sponsors as credible evidence—especially when the question is clear, the model is verified for purpose, and uncertainties are transparent. This article reviews real submissions where M&S supported dose selection, regimen bridging, DDI labeling, pediatric dosing, and systems-level benefit–risk insight. Contents Introduction Case 1 — Exposure Bridging (Pembrolizumab […]