Strategic Efficiency: How Model-Informed Drug Development (MIDD) Can Waive Clinical Studies and Accelerate Timelines
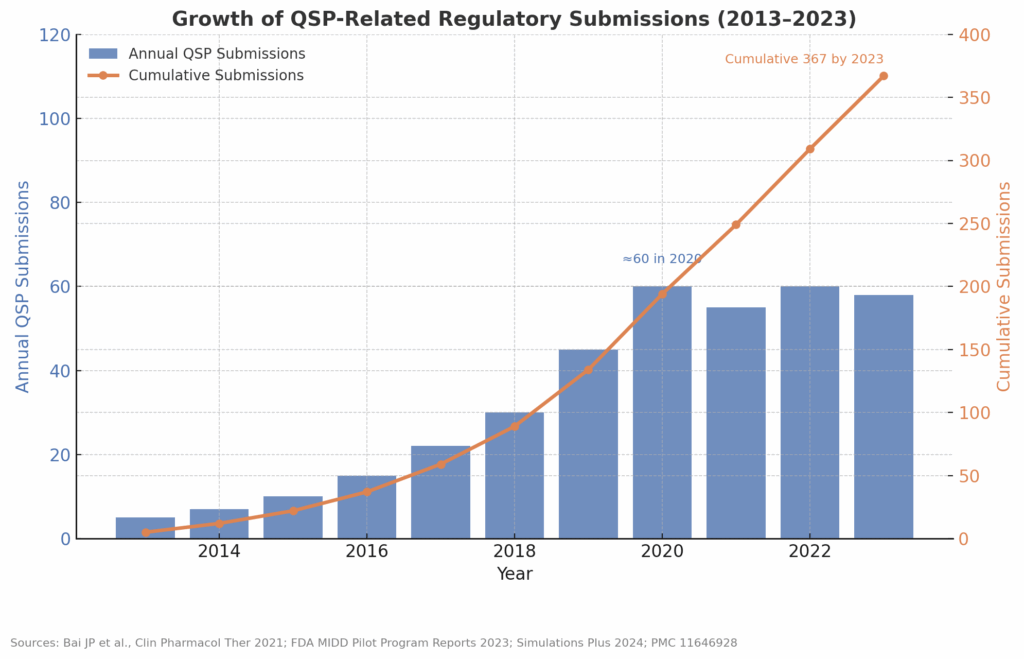
Introduction: The High Cost of “Checking the Box” For emerging biotech companies, cash runway is oxygen. The cost of developing a new drug has skyrocketed to over $2 billion, with the clinical phase consuming the vast majority of that budget. Every month spent in clinical development burns capital, shortens patent life, and delays the path […]