Executive Snapshot
- Purpose of 2.7.1: Turn biopharmaceutics and bioanalysis into decision-grade summaries that support clinical interpretation, bridging, and labeling. Keep it living from IND → EoP2 → NDA so the story is traceable and consistent across the dossier [1].
- Scope (in brief): BA/BE including food effect, dissolution/IVIVC/BCS, and clinical PK assay summaries (LLOQ/ULOQ, A/P, stability, ISR) with links to Module 5 for full reports [2][3][4][6][7].
- Relationship to other modules: 2.7.1 summarizes; 2.7.2 interprets dose/regimen; 2.3/Module 3 hold methods/specs; Module 5 holds full BE/FE and assay validation reports; the label reflects these conclusions [8].
What 2.7.1 Covers (and What It Does Not)
In scope (summaries):
- BA/BE results and conclusions including replicate/crossover for highly variable drugs as applicable [2][10].
- Food effect outcomes and dosing implications [5].
- Dissolution, IVIVC, and BCS considerations that underpin bridging or waivers [6][7].
- Bioanalytical method summaries for clinical assays: matrix, range, LLOQ/ULOQ, accuracy/precision, stability, ISR, and which studies used which assay/version [4].
Out of scope but must be cross referenced:
- Full BE and food effect reports → Module 5.3.1 [2][5].
- Full bioanalytical validation → Module 5.3.1.4 [4].
- Quality and dissolution methods/specifications → Module 3 (3.2.S/3.2.P) [6][7].
- Dose/regimen interpretation → 2.7.2 reference 2.7.1 only when assay or biopharm limits change PK interpretation [1][8].
Rule of thumb: 2.7.1 states what was measured and how it compares. 2.7.2 states what it means for dose and label.
Section by Section Blueprint (2.7.1.1 to 2.7.1.5)
2.7.1.1 Background and Biopharm Concepts
Briefly describe formulation(s), route, and biopharm risks for CPDS-1001 such as low solubility or pH-dependent dissolution. Declare up front where details live for example “Assay validation summaries are provided in 2.7.1.5; full reports are in 5.3.1.4.” Cite M4E(R2) for structure [1].
Submission style language:
“Two immediate release oral formulations capsule and tablet were used for CPDS-1001. Biopharmaceutic risks included low solubility and potential pH dependent dissolution. This section summarizes food effect, BA/BE, dissolution/IVIVC/BCS, and clinical bioanalytical methods. Full BE and assay validation reports are in Module 5; related methods/specifications are in Module 3.”
2.7.1.2 Clinical BA/BE food effect if stand alone
- Summarize designs, endpoints AUC and Cmax, test/reference, and the T/R ratios with 90% CIs against the 80 to 125% acceptance region [2][3].
- For food effect, state whether AUC or Cmax change is clinically meaningful tying to E–R or popPK where relevant [5][8].
- For modified release or highly variable drugs, note replicate/crossover and scaling approach [10].
Submission style language:
“Under fed conditions high fat AUC was 101% 90% CI 96 to 106 and Cmax 76% 90% CI 71 to 82 relative to fasted. As efficacy is AUC driven see 2.7.2 the lower Cmax is not clinically meaningful. Dosing with or without food is acceptable.” [5][8]
Submission style language:
“Comparative BA between the Phase 2 capsule and the to be marketed tablet for CPDS-1001 met BE. AUC 103% 90% CI 98 to 109 and Cmax 97% 90% CI 92 to 103. See 5.3.1.2 for the full report and 2.3 QOS for formulation details.” [2]

2.7.1.3 Comparative BA and Bridging Strategy
- Tie together formulation and process changes, scale up, and site transfers. Specify when you required BE vs comparative BA vs justification without study for example BCS waiver or IVIVC supported bridge [6][7].
- Provide a one page bridge table: change → evidence → conclusion → cross references.
Submission style language:
“A manufacturing site transfer occurred between Ph2 and Ph3. Batch characterization and biorelevant dissolution were comparable. A formal BE study was not required per risk assessment 2.3, 3.2.P.2 and 3.2.P.5. Population PK confirmed no meaningful formulation effect on exposure 2.7.2.” [6]
2.7.1.4 Dissolution, IVIVC, and BCS Considerations
Summarize dissolution methods and profiles with cross references to 3.2.P.5. Note whether IVIVC was established and whether a BCS based biowaiver ICH M9 is pursued [6][7].
Submission style language:
“Dissolution was discriminating across biorelevant media and predictive of in vivo rank order. An IVIVC was not established due to limited formulation variance see 3.2.P.5.” [6]
Submission style language:
“A BCS Class I waiver is supported by high solubility across pH 1 to 6.8, high permeability, and rapid dissolution at least 85% in 30 minutes for all strengths 3.2.P.2 and 3.2.P.5.” [7]
2.7.1.5 Bioanalytical Methods clinical PK assays
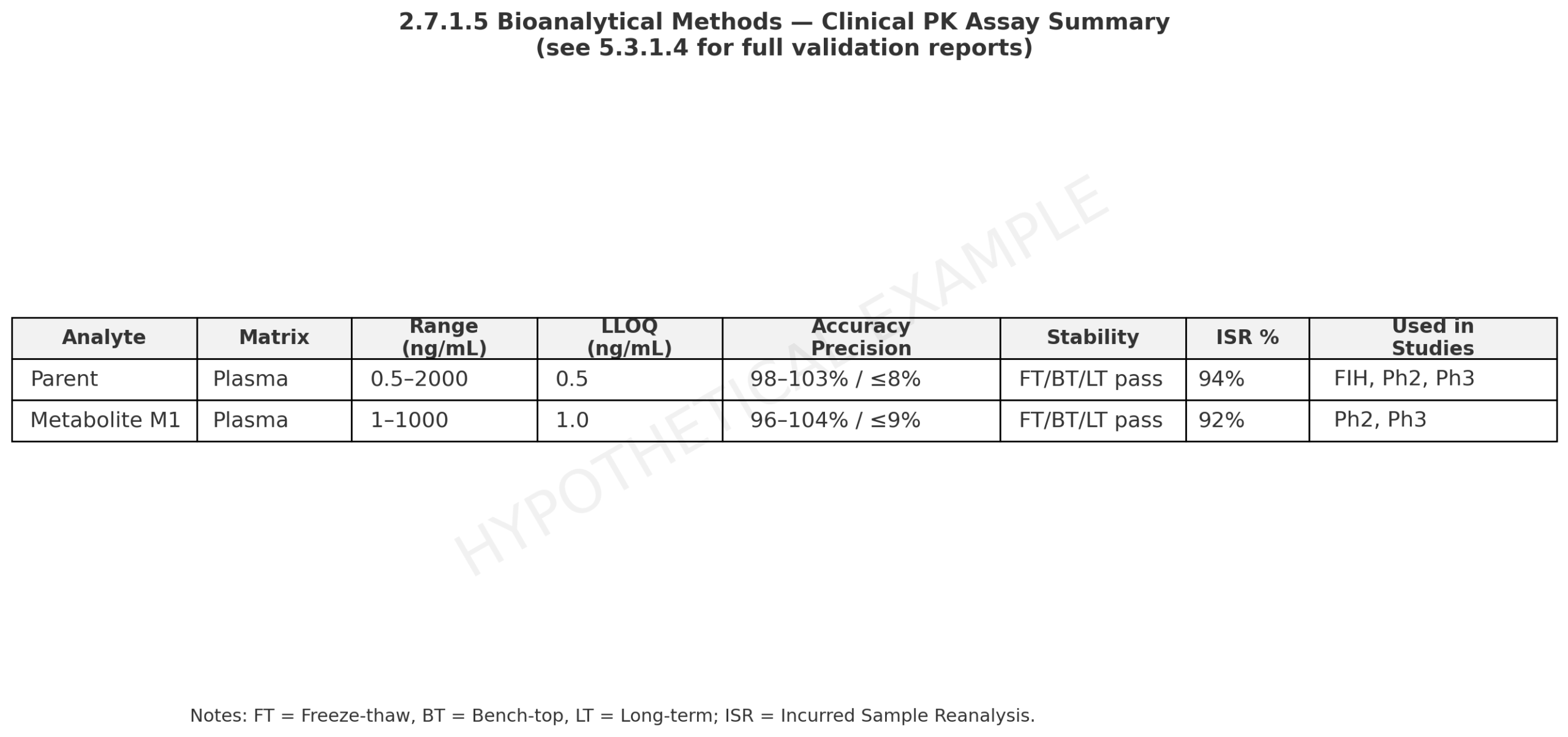
- Provide concise summaries of each assay used for clinical PK. Include matrix, range, LLOQ/ULOQ, accuracy/precision, stability, ISR, plus version history and which studies used which version [4].
- Avoid copying validation prose into 2.7.2. Reference 5.3.1.4 for full validation [4].
Submission style language:
“Plasma LC–MS/MS assay parent validated over 0.5 to 2,000 ng/mL LLOQ 0.5 ng/mL. Inter run accuracy 98 to 103% with precision ≤ 8%. Freeze thaw, bench top, and long term stability met acceptance. ISR pass rate 94%. See 5.3.1.4.” [4]
Submission style language:
“Assay v2 expanded the upper range before Ph3. Overlap and ISR confirmed no bias vs v1. Studies using each version are listed in Table X.” [4]
When to echo assay details in 2.7.2: only when they change PK interpretation for example trough near LLOQ [8].
How 2.7.1 Powers 2.7.2, QOS and Module 3, Module 5, and the Label
- 2.7.2: cites 2.7.1 when food effect or assay limits affect E–R conclusions and dose/regimen [5][8].
- 2.3 and Module 3: 2.7.1 bridges should align with 3.2.P.2/3.2.P.5 formulation/process and dissolution methods [6][7].
- Module 5: the source of truth for BE/FE and bioanalytical validation [2][4][5].
- Label Sections 2 and 12: language about with or without food, bioavailability, and comparability should map back to 2.7.1 evidence and be consistent with 2.7.2 [8].
Practical Examples What Good Looks Like
1) Food Effect That Does Not Change Dosing
- Result: Fed vs fasted AUC ~ 100%; Cmax ~ 75% below 80 to 125% for Cmax only.
- 2.7.1 write up: concise table of ratios and CI. One line: no clinically meaningful effect on overall exposure [5].
- 2.7.2 linkage: Efficacy is AUC driven. Safety E–R not Cmax driven → with or without food [8].
2) Early BE Failure, Late Stage Rescue
- Issue: First capsule to tablet BE failed on Cmax.
- Fix: Update dissolution method to better discriminate. Show alignment with in vivo rank order. Repeat BE passes.
- Documentation: 2.7.1.4 method change rationale plus QOS 2.3 and 3.2.P.5. Maintain 2.7.2 consistency [6].
3) Assay Upgrade Without PK Bias
IND to EoP2 to NDA: Keep 2.7.1 Living
IND: set up templates BA/BE, assay, dissolution/BCS, declare food effect and bridging strategies, and decide cross reference wording [1][2][4][5].
EoP2: populate FE and early BE or comparative BA results. Update dissolution/BCS status. Record ISR and versioning. Align with 2.7.2 and QOS 2.3 [6][7][8].
NDA: finalize all tables. Confirm traceability to 3.2.P and Module 5. Harmonize with proposed label [2][4][5][8].
Quick Authoring Templates
- BA/BE summary table 2.7.1.2: Study ID, design, T/R ratios with 90% CIs, BE conclusion [2][3].
- Bridge table 2.7.1.3: Change → evidence BE or comparative BA or IVIVC or BCS → conclusion → cross references [6][7].
- Assay table 2.7.1.5: Analyte, matrix, range, LLOQ/ULOQ, A/P, stability, ISR%, versioning, and which studies used each assay [4].
References
- ICH M4E(R2): The CTD — Efficacy (Module 2: Clinical Overview)
- FDA: Bioavailability and Bioequivalence Studies Submitted in NDAs or INDs — General Considerations
- EMA: Guideline on the Investigation of Bioequivalence
- FDA: Bioanalytical Method Validation — Guidance for Industry
- FDA: Assessing the Effects of Food on Drugs in INDs and NDAs — Clinical Pharmacology Considerations
- FDA: Extended Release Oral Dosage Forms — Development, Evaluation, and Application of IVIVC
- ICH M9: Biopharmaceutics Classification System Based Biowaivers
- FDA: Clinical Pharmacology Section of Labeling — Content and Format
- FDA: Physiologically Based Pharmacokinetic Analyses — Format and Content
- FDA: Bioequivalence Studies With PK Endpoints for Drugs Submitted Under an ANDA
Abbreviations
A/P Accuracy/Precision
AUC Area Under the Concentration–Time Curve
BA Bioavailability
BCS Biopharmaceutics Classification System
BE Bioequivalence
CI Confidence Interval
CSR Clinical Study Report
Cmax Maximum Observed Concentration
Ctrough Trough Concentration
DDI Drug–Drug Interaction
E–R Exposure–Response
IVIVC In Vitro/In Vivo Correlation
ISR Incurred Sample Reanalysis
LLOQ/ULOQ Lower/Upper Limit of Quantification
PBPK Physiologically Based Pharmacokinetics
popPK Population Pharmacokinetics
QOS Quality Overall Summary
QD Once Daily
T/R Test/Reference
t½ Terminal Half life