
Introduction: The High Cost of “Checking the Box”
For emerging biotech companies, cash runway is oxygen. The cost of developing a new drug has skyrocketed to over $2 billion, with the clinical phase consuming the vast majority of that budget. Every month spent in clinical development burns capital, shortens patent life, and delays the path to commercialization.
Historically, the industry approached Clinical Pharmacology as a rigid “check-the-box” exercise. If regulatory guidance mentioned a Drug-Drug Interaction (DDI) study, a Renal Impairment study, or a Food Effect study, the sponsor automatically commissioned a clinical trial to address it. This often resulted in “defensive development”—running trials not to answer scientific questions, but simply to satisfy a perceived regulatory requirement.
This traditional approach is inefficient, expensive, and increasingly unnecessary.
Today, Model-Informed Drug Development (MIDD) allows sponsors to simulate specific clinical scenarios in silico, often replacing the need for expensive, time-consuming in vivo studies. This is not merely academic theory; it is a regulatory-endorsed strategy with a proven Return on Investment (ROI).
A recent portfolio analysis by Pfizer (2025) demonstrated that the systematic application of MIDD yielded average savings of ~10 months of cycle time and $5 million per program [1]. For a small biotech, that time savings can be the difference between a successful exit and a critical funding gap.
What is MIDD?
MIDD involves the application of quantitative models to facilitate decision-making and regulatory submissions. It moves drug development from a trial-and-error approach to a predictive one.
- Physiologically-Based Pharmacokinetics (PBPK): Simulates the drug’s behavior based on human physiology (blood flow, organ size) and drug properties (lipophilicity, metabolism). It is the “gold standard” for predicting drug interactions.
- Population Pharmacokinetics (PopPK): Analyzes data from all patients in a trial simultaneously to identify sources of variability (age, weight, kidney function) that affect drug exposure.
- Exposure-Response (E-R) Modeling: Links the concentration of the drug in the blood to the clinical outcomes (efficacy and safety), helping to define the “sweet spot” for dosing.
Since the inclusion of MIDD in the PDUFA VI commitments, the FDA has actively encouraged sponsors to use modeling to address key labeling questions [2]. In fact, over 25% of all new drug approvals now contain PBPK modeling as pivotal evidence to support dosing recommendations.
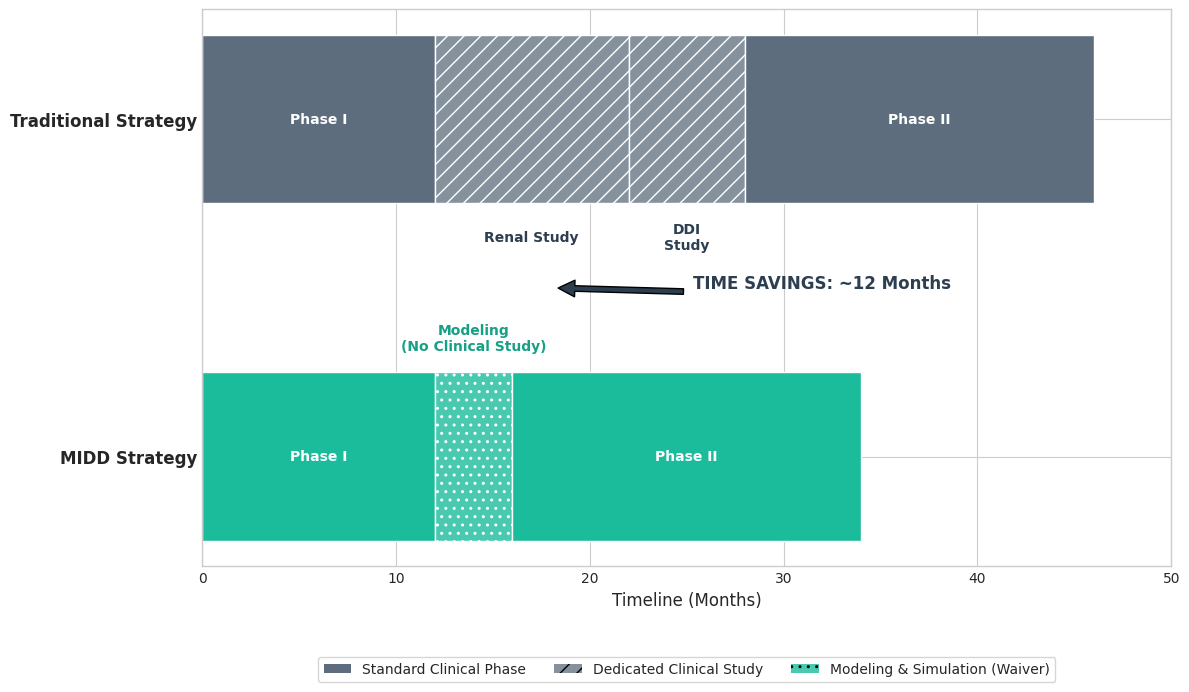
Practical Application 1: Waiving Drug-Drug Interaction (DDI) Studies
One of the most common bottlenecks in Phase II/III is the requirement to characterize how a drug interacts with “perpetrator” agents. If your drug is metabolized by the CYP3A4 enzyme, regulators need to know: What happens if a patient takes it with a strong inhibitor (like ketoconazole) or a moderate inhibitor (like diltiazem)?
The Traditional Cost:
Conducting a dedicated clinical DDI study is a massive logistical undertaking. It typically costs $500k–$1M and takes 6–9 months to design, recruit healthy volunteers, and execute. Furthermore, testing multiple categories of inhibitors (strong, moderate, weak) requires multiple arms or multiple studies.
The MIDD Solution:
If you have high-quality in vitro data (microsomal stability, inhibition constants), a PBPK model can be built to simulate these interactions. The strategy involves verifying the model against one “worst-case” clinical scenario (e.g., a strong inhibitor). Once validated, the FDA often permits the use of the model to predict the effects of “moderate” or “weak” inhibitors without running further trials.
Deep Dive Case Study: Palbociclib (Ibrance)
Pfizer’s development of palbociclib, a CDK4/6 inhibitor for breast cancer, serves as a landmark example. The drug is a CYP3A substrate.
- The Challenge: Assessing the safety of the drug when co-administered with a wide range of common medications (antibiotics, antifungals, heart medications) that inhibit or induce CYP3A.
- The Model: Pfizer developed a PBPK model and verified it using clinical data from a study with itraconazole (a strong inhibitor) and rifampin (a strong inducer).
- The Win: Instead of running additional clinical trials for moderate inhibitors (like diltiazem) or moderate inducers (like efavirenz), they simulated these scenarios.
- Outcome: The FDA approved specific dosing instructions in the label based solely on these simulations. For example, the label advises reducing the dose to 75 mg when taken with moderate inhibitors—a recommendation derived entirely from PBPK, saving millions in clinical costs [3].
Table 1: ROI Analysis of DDI Assessment (Clinical Trial vs. MIDD)
| Metric | Traditional Clinical DDI Study | MIDD Strategy (PBPK Modeling) | Strategic Advantage |
|---|---|---|---|
| Direct Cost | $500,000 – $1,000,000+ (per study) | $50,000 – $150,000 | ~90% Cost Reduction |
| Timeline | 6 – 12 Months | 2 – 3 Months | ~75% Time Savings |
| Operational Risk | High (Recruitment delays, AEs) | Low (In silico simulation only) | Zero Safety Risk |
| Scalability | Linear | Exponential | Comprehensive Label |
| Deliverable | Clinical Study Report (CSR) | PBPK Simulation Report | Accepted for Labeling |
Practical Application 2: Waiving Organ Impairment Studies
Regulators require knowledge of how a drug is cleared by patients with compromised organ function. However, recruiting patients with “Severe Renal Impairment” is notoriously difficult. These patients are often elderly, have multiple comorbidities, and are reluctant to enroll in non-therapeutic trials.
The Traditional Cost:
A standalone renal impairment study often becomes the “critical path” delay. If recruitment stalls, it can delay the NDA filing by 12 months or more.
The MIDD Solution:
Instead of a standalone study, sponsors can use a Population PK (PopPK) analysis. By collecting sparse PK samples from the hundreds of patients already enrolled in the pivotal Phase II/III trials, the model can assess renal function as a “covariate.” Since many cancer or chronic disease patients naturally have mild-to-moderate impairment, this real-world data is often sufficient.
Deep Dive Case Study: Vismodegib (Erivedge)
Vismodegib is a hedgehog pathway inhibitor indicated for basal cell carcinoma.
- The Challenge: The drug is eliminated primarily by metabolism and excretion, but regulators needed assurance that renal impairment wouldn’t cause dangerous accumulation.
- The Strategy: The sponsor did not run a dedicated renal impairment study. Instead, they conducted a PopPK analysis using data from patients in the pivotal trial.
- The Analysis: The model demonstrated that creatinine clearance (a marker of kidney function) was not a significant covariate—meaning drug levels were comparable between patients with normal function and those with impairment.
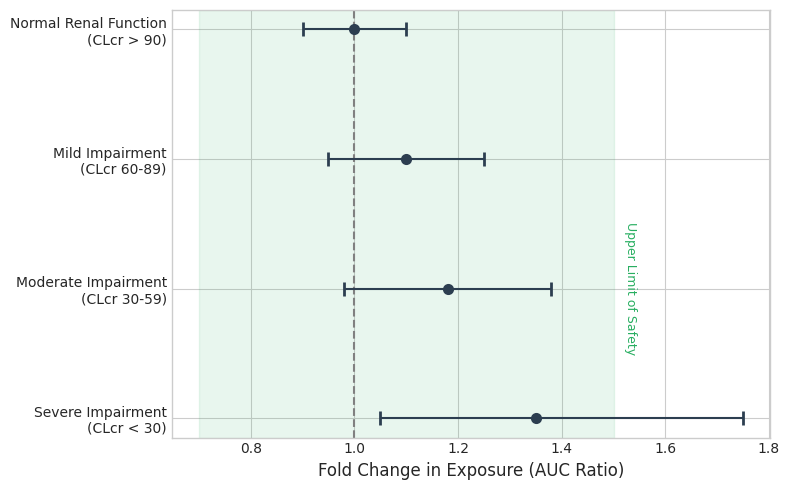
Decoding the Data: Figure 2 (above) illustrates this concept. The green shaded area represents the “Safety Window” (typically 80-125% of standard exposure). The bars represent the drug exposure in patients with mild and moderate kidney damage. Because these bars fall almost entirely within the green zone, the model proves that drug levels do not spike dangerously high.
Outcome: The FDA accepted this analysis to support the label statement: “No dose adjustment is required for patients with mild to moderate renal impairment,” effectively waiving the need for a dedicated study [4].
Practical Application 3: Dose Optimization & Flat Dosing
The FDA’s “Project Optimus” initiative has shifted the paradigm from finding the Maximum Tolerated Dose (MTD) to identifying the Optimal Biological Dose. Furthermore, operationally, hospitals and pharmacies prefer “flat dosing” (e.g., 480 mg fixed) over weight-based dosing (e.g., 3 mg/kg) to reduce calculation errors and drug waste.
The Traditional Cost:
Running a new, randomized Phase III trial to prove that a flat dose is statistically equivalent to a weight-based dose is prohibitively expensive and ethically questionable.
The MIDD Solution:
Exposure-Response (E-R) modeling can bridge the gap. By simulating exposures across thousands of virtual patients with varying body weights, sponsors can demonstrate that a flat dose maintains efficacy in heavy patients while maintaining safety in light patients.
Deep Dive Case Study: Nivolumab (Opdivo)
Bristol Myers Squibb sought to optimize the dosing of nivolumab to improve convenience. The original approval was for 3 mg/kg every 2 weeks (Q2W). They wanted to switch to a flat dose of 480 mg every 4 weeks (Q4W).
- The Analysis: They simulated the pharmacokinetic profile of the new regimen.

Decoding the Data: As shown in Figure 3, the flat dose (purple dot) results in a higher peak concentration (Cmax) in lighter patients compared to the weight-based dose (blue dot). However, the “Safety” curve (Red) is flat at these concentrations—meaning the risk of toxicity does not increase significantly despite the higher peak. Simultaneously, the efficacy (Green curve) remains on the plateau.
Outcome: This “Safety Margin” argument allowed the FDA to approve the new dosing regimen across multiple indications based primarily on modeling data, without a new efficacy trial [5].
Practical Application 4: Bispecific Antibodies (Step-Up Dosing)
MIDD is critical for complex modalities where clinical trials are unsafe or difficult to design. Bispecific antibodies (e.g., T-cell engagers) often cause Cytokine Release Syndrome (CRS) upon the first dose. To mitigate this, complex “step-up” priming doses are used.
Deep Dive Case Study: Teclistamab (Tecvayli)
Teclistamab is a BCMA x CD3 bispecific antibody for multiple myeloma.
- The Challenge: The approved weekly dosing schedule was burdensome for patients and required frequent hospital visits. The sponsor wanted to allow patients who achieved a complete response to switch to a less frequent Every-2-Week (Q2W) schedule.
- The Solution: Rather than running a new randomized trial to test Q2W dosing, they used Exposure-Response modeling from the MajesTEC-1 study. The model predicted that even with the lower trough concentrations from Q2W dosing, the drug levels would remain well above the target threshold for efficacy.
- Outcome: The FDA approved the switch to Q2W dosing for responders based on the modeling data. This “patient-centric” update gave the drug a significant commercial advantage by reducing the burden on patients [6].
Practical Application 5: Antibody-Drug Conjugates (Payload DDI)
Antibody-Drug Conjugates (ADCs) present a unique regulatory puzzle. They consist of a large antibody (biologic) and a toxic small-molecule payload. Regulators worry: Will the toxic payload accumulate if the patient takes a CYP inhibitor?
Deep Dive Case Study: Polatuzumab vedotin (Polivy)
Polatuzumab vedotin is an ADC used for Diffuse Large B-Cell Lymphoma. Its payload, MMAE, is a substrate of CYP3A4.
- The Challenge: The FDA needed to know if strong CYP3A inhibitors would increase MMAE exposure to toxic levels.
- The Solution: The sponsor developed a PBPK model that accounted for the release of MMAE from the antibody and its subsequent metabolism. They simulated the interaction with strong CYP3A inhibitors (like ketoconazole) and inducers (like rifampin).
- Outcome: The model predicted that interactions would be limited. The FDA granted a label claim describing the interaction risk (or lack thereof) based on the PBPK report, waiving the need for a dedicated clinical DDI study for the payload [7].
The Strategic Checklist: Is Your Program Ready?
MIDD is most effective when integrated early. Waiting until the Pre-NDA meeting to propose a waiver is often too late—the FDA requires time to review and accept the modeling plan.
Table 2: Strategic Readiness Checklist for MIDD
| Strategic Component | Key Question for the Sponsor | Why it Matters? |
|---|---|---|
| 1. In Vitro Data Quality | Do you have high-quality metabolic stability and inhibition constants (Ki, IC50) for your drug? | High-quality in vitro inputs are the “fuel” for PBPK models. Regulators will reject models based on poor data. |
| 2. Phase I Sampling | Did you collect adequate PK samples (sparse or rich) in your First-in-Human study? | These clinical samples are required to verify the baseline model before simulating more complex scenarios (like DDIs). |
| 3. Regulatory Timing | Have you proposed a “modeling & waiver strategy” in your Pre-IND or End-of-Phase 2 meeting? | Regulators need to agree to the plan before you submit the results. Early alignment prevents last-minute study demands. |
| 4. Critical Path Analysis | Is your NDA filing timeline dependent on the results of a specific clinical study (e.g., DDI or Renal)? | If a study is on the critical path, modeling offers the highest ROI by removing the risk of clinical delays. |
Conclusion
Clinical Pharmacology is no longer about generating data for data’s sake. It is a strategic lever that, when pulled correctly, can compress timelines and preserve capital.
By replacing “check-box” clinical studies with regulatory-grade modeling, ClinPharm Dev Solutions helps partners navigate the path to approval more efficiently. Don’t wait for the FDA to ask for a study—partner with us to build your waiver strategy today.
Abbreviations
- ADC: Antibody-Drug Conjugate
- CRS: Cytokine Release Syndrome
- CYP: Cytochrome P450
- DDI: Drug-Drug Interaction
- E-R: Exposure-Response
- FDA: Food and Drug Administration
- IND: Investigational New Drug
- MIDD: Model-Informed Drug Development
- MMAE: Monomethyl auristatin E
- MTD: Maximum Tolerated Dose
- NDA: New Drug Application
- PBPK: Physiologically-Based Pharmacokinetics
- PDUFA: Prescription Drug User Fee Act
- PopPK: Population Pharmacokinetics
- Q2W: Every 2 Weeks
- Q4W: Every 4 Weeks
- ROI: Return on Investment
References
- Pfizer Analysis (2025). Impact of Model-Informed Drug Development on Cycle Times and Costs. Clinical Pharmacology & Therapeutics.
- U.S. Food and Drug Administration (2017). Prescription Drug User Fee Act (PDUFA) VI.
- Yu, Y. et al. (2017). Physiologically Based Pharmacokinetic Modeling of Palbociclib. Journal of Clinical Pharmacology.
- FDA Clinical Pharmacology Review: Vismodegib (NDA 203388).
- Long, G. et al. (2018). Nivolumab Dosing: Flat Dose vs Weight-Based. Cancer Chemotherapy and Pharmacology.
- MajesTEC-1 Study Group (2022). Teclistamab in Relapsed or Refractory Multiple Myeloma. NEJM.
- Li, C. et al. (2020). PBPK Modeling for Polatuzumab Vedotin: Label for DDIs Without Dedicated Trials. CPT: Pharmacometrics & Systems Pharmacology.