Introduction
Oncology drug development has traditionally shunned healthy volunteer (HV) participation due to the high toxicity of classic chemotherapies [1]. Cytotoxic agents non-selectively kill rapidly dividing cells, causing severe side effects that would be unethical in individuals without cancer. However, the therapeutic landscape is changing. Over the past two decades, targeted therapies—drugs aimed at specific molecular pathways (e.g., kinase inhibitors)—and immunotherapies have emerged as less toxic alternatives [2]. These agents often spare normal cells and produce more tolerable side effect profiles compared to conventional chemotherapy. Consequently, there is growing acceptance of limited clinical studies in normal healthy volunteers (NHVs) for certain oncology drugs, especially in early clinical pharmacology trials [1].
This shift is fueled by both scientific and practical motivations. Targeted oncology drugs are generally non-genotoxic and do not cause irreversible damage like classic chemotherapy [3]. Regulators now acknowledge that the favorable safety profiles of some targeted agents may justify NHV trials under specific conditions [3]. When thoughtfully applied, HV studies can accelerate timelines, yield cleaner pharmacology data, and reduce ethical concerns around enrolling patients into subtherapeutic dose cohorts.
Regulatory Guidance Landscape
The inclusion of healthy volunteers in oncology trials has historically been limited by ethical and safety concerns. However, evolving guidance from global regulatory authorities now provides a structured pathway for incorporating HV studies into oncology development—especially for non-cytotoxic agents.
The European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA) both acknowledge that early clinical pharmacology studies in HVs may be appropriate when sufficient preclinical safety data exist. The EMA’s guideline on strategies to identify and mitigate risks in first-in-human and early trials (EMA/CHMP/SWP/28367/07 Rev. 1) outlines a risk-based approach, assessing genotoxic potential, off-target effects, and predicted systemic exposures. Similarly, the FDA’s oncology-specific guidance highlights HV suitability for label-enabling studies like food-effect, QTc, and drug-drug interaction evaluations.
Together, these guidelines support HV studies as part of a holistic clinical development strategy—provided robust safety justifications, multidisciplinary sign-offs, and contingency planning are in place.
Why Use Healthy Volunteers in Oncology?
There are several advantages to using healthy volunteers in early oncology development:
- Faster Recruitment & Lower Cost: HV studies are easier to recruit, quicker to execute, and less expensive than oncology patient studies [2].
- Cleaner PK/PD Data: HVs have minimal comorbidities and no concomitant medications, resulting in more interpretable data [2].
- Ethical Allocation of Patient Participation: Conducting non-therapeutic trials in HVs helps avoid exposing cancer patients to low or subtherapeutic doses [4].
- Rich Sampling and Monitoring: HV studies can accommodate intensive blood sampling, confinement, and biomarker evaluations.
- Label-Enabling Evidence: Studies such as QTc, food-effect, mass balance, and DDI evaluations are often better suited for HVs [5].
What Types of Trials Can Involve Healthy Volunteers?
Below is a summary of clinical pharmacology studies commonly conducted in HVs that support oncology development:
| Study Type | Typical HV Design | Purpose |
|---|---|---|
| Phase 0 / Microdosing | Sub-therapeutic single dose | Early human PK insight [6] |
| Human ADME / Mass Balance | Radiolabeled single dose | Characterize absorption, metabolism, excretion |
| Food Effect | Fed vs. fasted crossover | Labeling: guidance on meal timing [8] |
| Drug-Drug Interaction | With/without strong perpetrator | Evaluate metabolic/transporter risks |
| Bioavailability / Bioequivalence | Two formulations, crossover | Compare tablet vs capsule, or generics |
| QTc / Cardiac Safety | Single or supra-therapeutic dose | Measure QT interval effect |
| Special Population PK | Renal-impaired, Japanese, etc. | Ethnic or impairment-based dose adjustment |
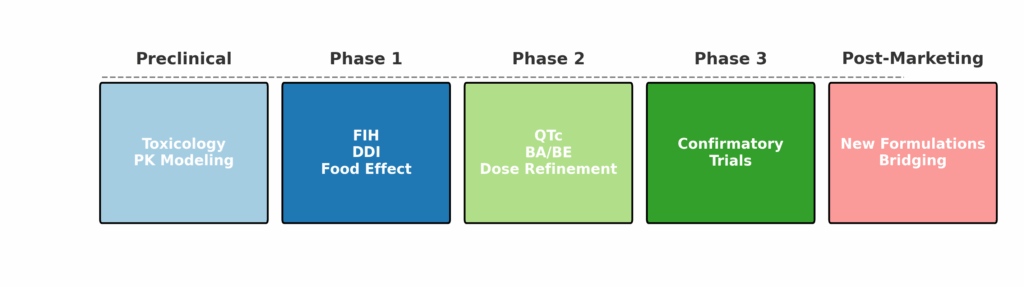
When Are HV Studies Conducted in Oncology?
Phase 0–1: PK/PD, food-effect, DDI studies
Phase 2–3: QTc, mass balance, BA/BE
Post-marketing: Bridging and new formulations
Decision Framework for Including Healthy Volunteers
Before initiating HV studies, a cross-functional review should confirm:
- Is the compound non-cytotoxic?
- Is there no significant genotoxicity risk?
- Can a safe starting dose be defined?
- Does the study lack therapeutic intent?
Case Examples
Case 1 – ETC-206 (MNK 1/2 inhibitor)
ETC-206 is a selective kinase inhibitor developed for cancer treatment. A first-in-human trial in healthy male volunteers in Singapore showed that single oral doses of 10 mg and 20 mg were well tolerated [7]. No serious adverse events were observed, and investigators were able to measure both pharmacokinetics and pharmacodynamic markers—specifically, phosphorylation of eIF4E. The study supported a pharmacologically active and safe dose range for future patient trials.
Case 2 – Lazertinib (EGFR TKI)
Lazertinib is a third-generation EGFR tyrosine kinase inhibitor developed for NSCLC. A crossover study in 24 HVs (12 Asian and 12 non-Asian) examined the impact of food and ethnicity on lazertinib PK [8]. The study found:
- A <15% increase in AUC with a high-fat meal (not clinically significant)
- Slightly higher systemic exposure in Asians, not requiring dose adjustment
The data informed the labeling recommendation: “May be taken with or without food.”
Conclusion
Healthy volunteer studies are emerging as a smart, ethical, and efficient tool in oncology drug development—especially for targeted, non-cytotoxic agents. These studies allow:
- Faster and cleaner PK/PD insights
- Reduced development timelines
- Safer patient-centric dose escalation
- Ethical avoidance of low-efficacy patient cohorts
While not suitable for all molecules (e.g., cytotoxics or aggressive immunotherapies), HV trials have proven safe and useful for a wide range of small molecules and selected biologics. With robust safety data and thoughtful study design, HV inclusion is increasingly becoming a strategic decision for biotech sponsors.
References
- Ahmed S, Haigentz M, Miller VA, et al. The Evolving Role of Healthy Volunteers in Oncology Drug Development. Clin Pharmacol Ther. 2020;107(1):112–118.
- Paglialunga S, van Haarst AD. Advantages and challenges of using healthy volunteer studies in oncology drug development. Br J Clin Pharmacol. 2024;90(3):551–560.
- de Las Heras B, Martin M, Solans BP, et al. Regulatory and safety considerations for first-in-human studies in healthy volunteers for oncology drugs. Ther Innov Regul Sci. 2022;56(5):879–887.
- Radanovic A, Feller T, Gatschelhofer C, et al. Safety and tolerability of immuno-oncology therapies in healthy volunteers: a review of 38 clinical trials. Front Pharmacol. 2022;13:982934.
- Vlachogiannis G, Pisal N, Kaplan S, et al. Implementation of healthy volunteer studies in early-phase oncology: A multidisciplinary risk assessment approach. J Clin Pharmacol. 2023;63(4):423–432.
- de Vries Schultink AHM, Dorlo TPC, Deliens L, et al. Integration of Phase 0 microdosing and first-in-human trials in oncology: rationale and case studies. Eur J Cancer. 2019;110:123–131.
- Seo JH, Kim SH, Shin JS, et al. A phase I, randomized, double-blind, placebo-controlled study to assess the pharmacokinetics and pharmacodynamics of ETC-206 in healthy male volunteers. Cancer Chemother Pharmacol. 2021;87(1):133–142.
- Lee SH, Cho YB, Kim JW, et al. Food effect and ethnic sensitivity study of lazertinib in healthy volunteers: implications for global development. Br J Clin Pharmacol. 2023;89(1):88–96.
- EMA. Guideline on strategies to identify and mitigate risks for first-in-human and early clinical trials with investigational medicinal products. EMA/CHMP/SWP/28367/07 Rev. 1, 2017.
- FDA. Cancer Clinical Trial Eligibility Criteria: Patients with Organ Dysfunction or Prior or Concurrent Malignancies. Guidance for Industry. March 2020.