Even if you never build a PBPK model in-house, an early decision framework—knowing when PBPK can replace, complement, or simply de-risk clinical studies—cuts duplicated work and answers “what-if” regulatory questions before they become clock-stopping RFIs.
Why deciding early—not necessarily modeling early—saves studies, budget, and review stress
Small molecule therapies—and, occasionally, intravenous antibody–drug conjugates (ADCs) carrying small‑molecule payloads—face a crowded landscape of potential drug–drug interactions (DDIs). Classic perpetrators such as azole antifungals or proton‑pump inhibitors can double systemic exposure, and combination regimens mean few investigational drugs travel alone.
Regulators no longer expect you to solve every interaction question before first in human studies, but they do want clear evidence that you’ve thought through the sequence: Which risks can be screened clinically, which can be simulated mechanistically, and which can be postponed until confirmatory phases if early signals stay benign. A concise PBPK decision framework—mapped out during discovery and revisited at each development milestone—helps you budget intelligently, avoid late‑phase study surprises, and walk into FDA or EMA meetings with a staged plan instead of open‑ended promises.
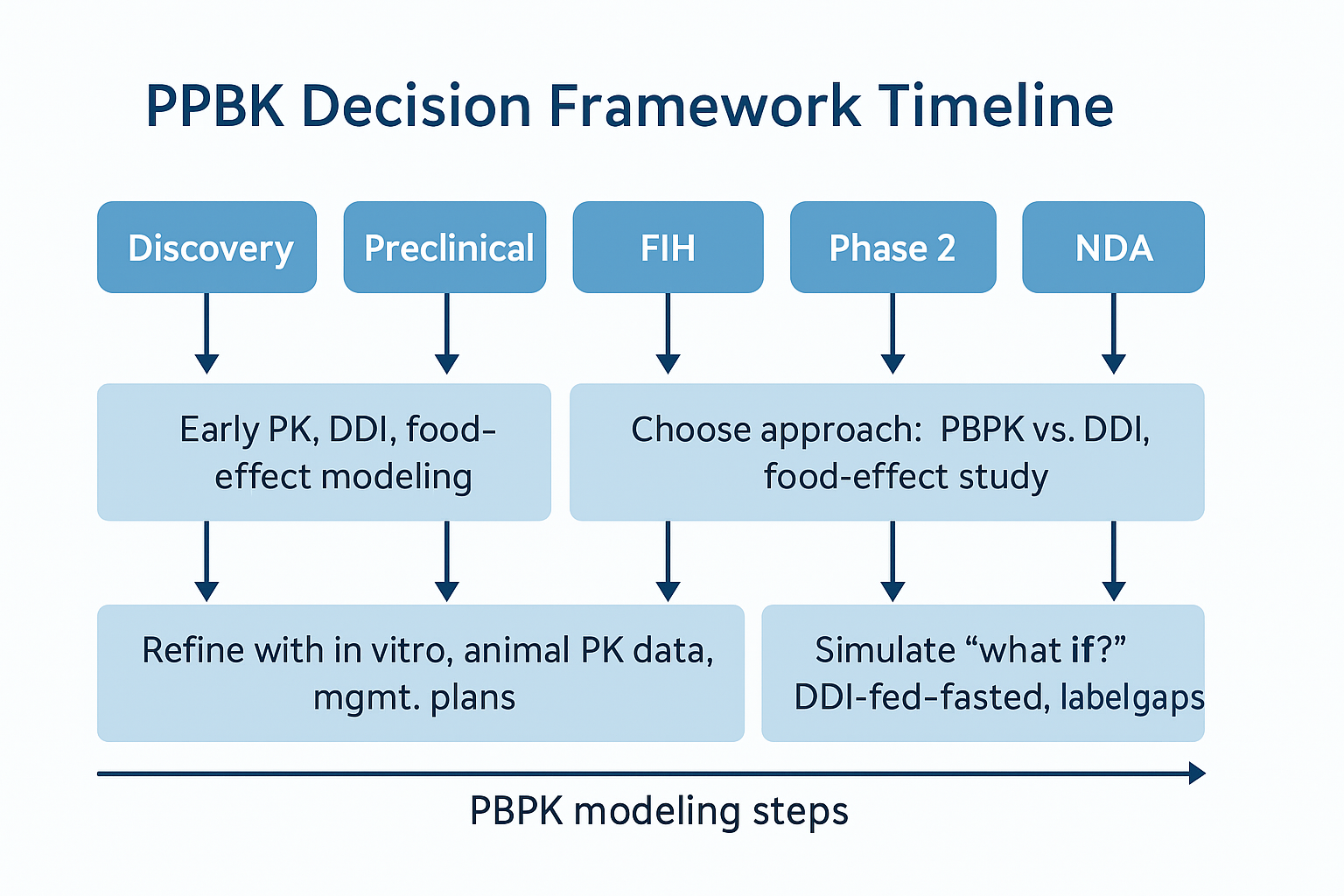
Figure 1. Decide early: minimal anchor studies feed outsourced PBPK simulations, closing DDI & food-effect gaps before NDA.
1 Why PBPK belongs on your critical path
Before diving into the evidence, here are three signals that make a proactive PBPK stance hard to ignore:
• Guidances are clear. FDA’s PBPK format guide [1], FDA’s in‑vitro drug–interaction guidance (2020) [3], and EMA’s PBPK reporting template [2] spell out exactly how reviewers assess models; ICH M12 (2024) explicitly lists PBPK as an accepted tool for DDI risk mitigation [4].
• It’s in the approval stats. Roughly a quarter of EMA marketing-authorisation dossiers in 2022–23 cited PBPK to defend DDI, food-effect, or special-population dosing [8].
• RFIs are tougher. The FDA–AAPS PBPK workshop in October 2023 [7] highlighted how recent FDA review cycles ask sponsors to simulate strong‑inducer scenarios within seven days—impossible without a PBPK plan already in place.
Even if modeling is outsourced, deciding whether PBPK will be required—and pencilling in budget, timelines, and CRO partners—belongs in discovery, not after first-in-human (FIH) data arrive.
2 PBPK in DDI strategy—where it pays off most
| Regulatory question | All-clinical fix | Lean fix with PBPK guidance |
|---|---|---|
| Victim (CYP3A4 substrate) | 3-arm ketoconazole ± rifampin | 1 ketoconazole anchor → PBPK simulates moderate/weak inhibitors & inducers |
| Perpetrator (induction) | Two midazolam studies | Micro-dose midazolam anchor → PBPK predicts AUCR grid |
| Transporter risk (OATP/BCRP) | Full rosuvastatin study | PBPK simulates statin risk; run study only if AUCR > 1.25 × |
Our advisory deliverable: a decision tree that maps your in‑vitro data to current FDA/EMA guidance (including the 2020 FDA in‑vitro DDI guide [3]) and shows exactly which anchors unlock PBPK substitutions.
That single visual arms your team with a regulator‑aligned roadmap for trimming unnecessary studies while avoiding review delays.
3 PBPK for food-effect predictions
Note: The fed–fasted considerations in this section apply only to orally administered small molecules; intravenously delivered ADCs are exempt from food-effect studies.
PBPK models can de-risk early formulation choices and limit the size of your fed–fasted comparison.
- BCS Class I/III drugs with fast dissolution can often skip food-effect studies altogether if PBPK + in-vitro data show no meaningful exposure shift.
- BCS Class II/IV drugs with pH-dependent solubility can use PBPK to justify a narrower fed/fasted study or to preempt a food-labeling issue at NDA.
Sponsors who model early avoid the common trap: finding a significant food effect at the pivotal stage, then rushing to reformulate.
4 When to act—even if you don’t model in-house
You don’t need to build models yourself. But you do need to identify which studies PBPK can replace or shrink—before your timeline locks up.
Figure 1 (above) gives a rough timeline: the earlier you feed in-vitro and anchor data to your modeling partner, the sooner they can simulate what’s skippable.
We help small and midsize sponsors:
- Make early-phase go/no-go PBPK decisions based on study feasibility and exposure margin
- Align your plan to FDA and EMA expectations before submission
- Identify CROs or modeling consultants who can execute efficiently
References
[1] U.S. FDA. Physiologically Based Pharmacokinetic Analyses — Format and Content. 2018.
[2] European Medicines Agency. Guideline on the qualification and reporting of PBPK modelling and simulation. 2018.
[3] U.S. FDA. In Vitro Drug Interaction Studies — Cytochrome P450 Enzyme- and Transporter-Mediated Interactions. 2020.
[4] ICH M12: Drug Interaction Studies, Step 4 Guideline. 2024.
[5] U.S. FDA. Clinical Drug Interaction Studies With Therapeutic Proteins. Draft Guidance. 2023.
[6] Kuemmel et al. PBPK submissions to the U.S. FDA: An update on applications and acceptance. CPT Pharmacometrics Syst Pharmacol. 2019.
[7] FDA-AAPS Workshop on PBPK Modeling and Simulation for Regulatory Submissions. October 2023.
[8] EMA Human Medicines Highlights 2022–2023: Public assessment reports and marketing-authorisation data.
