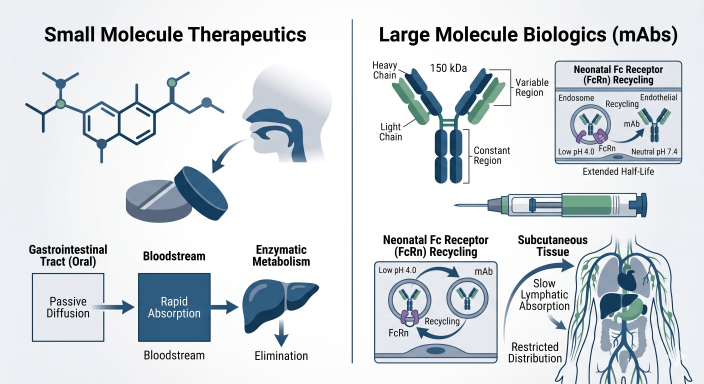
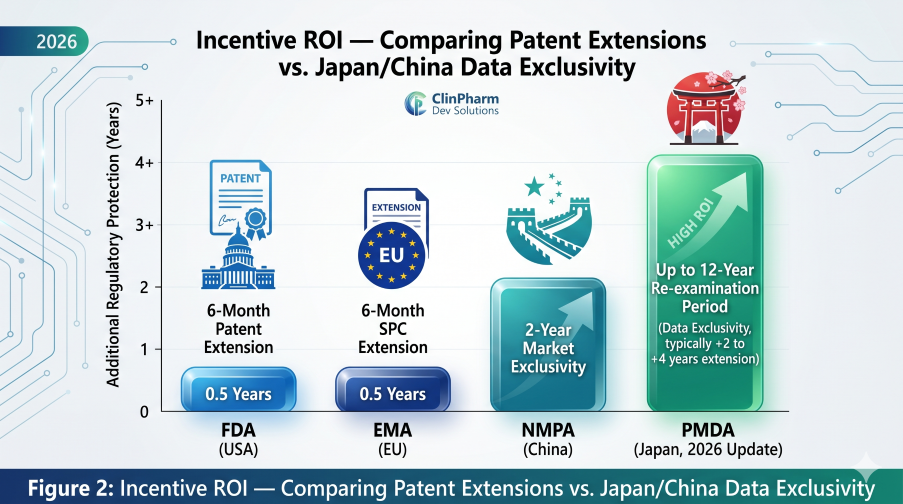
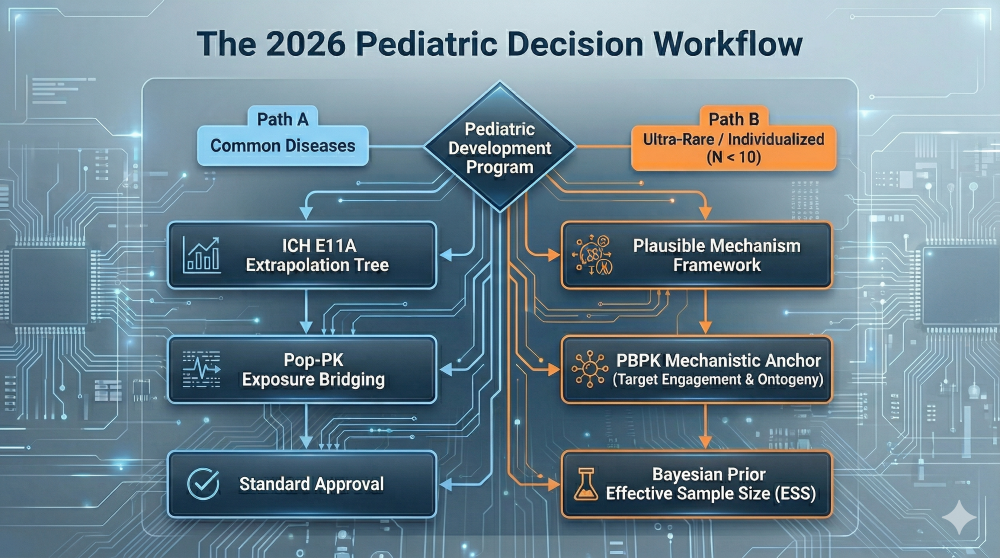
Comprehensive Clinical Pharmacology and QCP: Modality-Specific Requirements in Drug Development
Executive Outline: Series on Modality-Specific Clinical Pharmacology To address the rapidly evolving landscape of therapeutic modalities and the increasing complexity of regulatory requirements, this comprehensive report serves as the foundational document in a specialized series detailing modality-specific requirements for clinical pharmacology, drug metabolism and pharmacokinetics (DMPK), and pharmacometrics. The series is structured to provide an […]