IND to NDA Insights for Biotech Success
Practical guidance, real examples, and startup-focused strategies — from IND through NDA and beyond.
The Dossier Deep Dive series is designed for emerging biopharma and biotech companies navigating the complex journey from first-in-human studies to marketing authorization.
Each article breaks down a specific section of the Common Technical Document (CTD), explaining its purpose, regulatory expectations, and how early strategic alignment can save months in development timelines.
Drawing from real-world case studies, hands-on clinical pharmacology expertise, and regulatory experience across FDA and EMA submissions, this series equips your team with actionable insights to avoid common pitfalls, maximize efficiency, and prepare for global success.
What you will learn in This Series
- How each CTD section contributes to regulatory approval — and why early alignment matters.
- Key differences between IND, NDA, and IMPD submissions and how to prepare for multiple regions at once.
- Practical tips for organizing and maintaining “living” CTD modules.
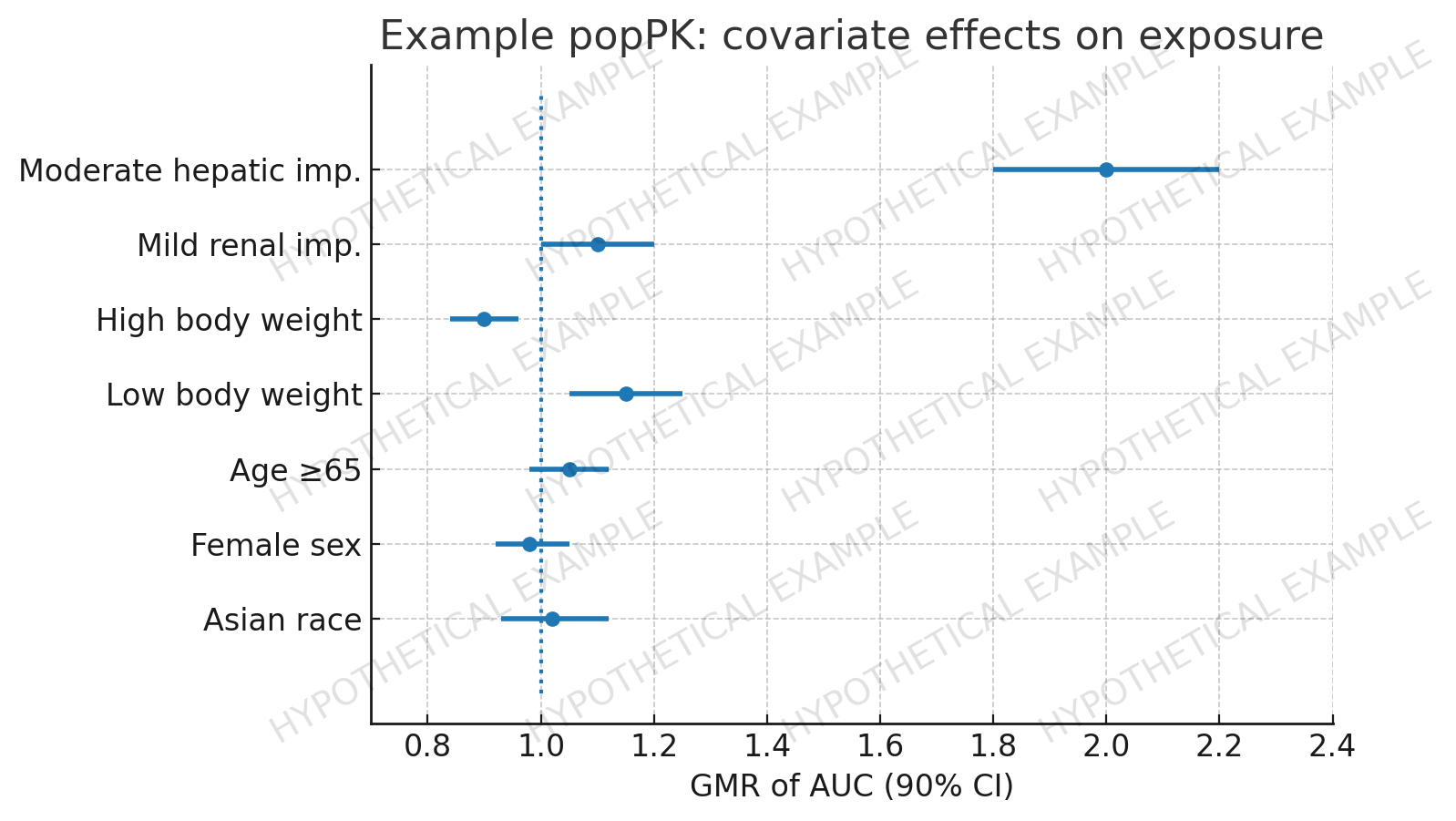
- Guidance on what is required in each CTD module for different therapeutic modalities (e.g., small molecules, biologics, ADCs, radiopharmaceuticals, RNA therapies) and disease areas (e.g., oncology, rare diseases, immunology).
- Real-world examples of how startups have saved 3–6 months through smart dossier planning.