From Bioanalysis to Biowaivers: Writing Module 2.7.1 That Travels From IND to NDA
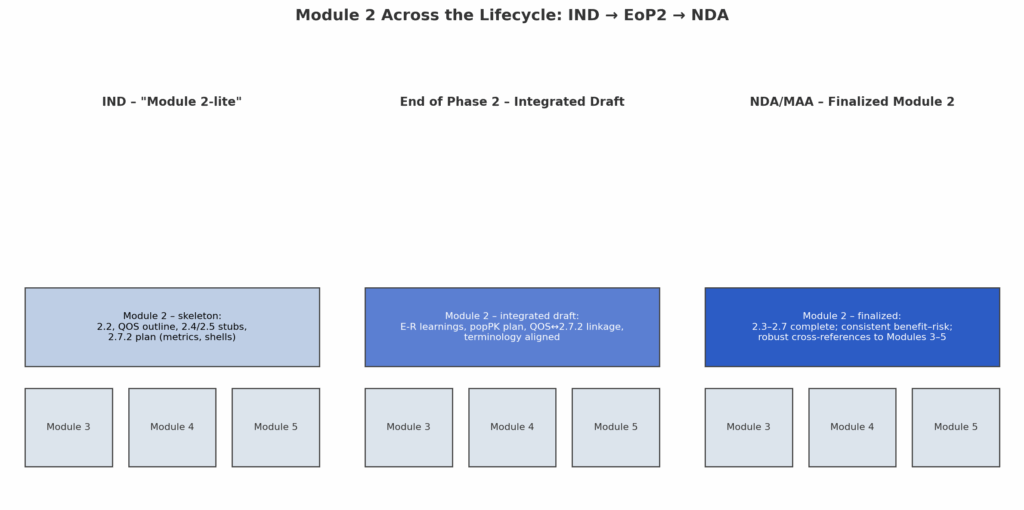
Executive Snapshot Purpose of 2.7.1: Turn biopharmaceutics and bioanalysis into decision-grade summaries that support clinical interpretation, bridging, and labeling. Keep it living from IND → EoP2 → NDA so the story is traceable and consistent across the dossier [1]. Scope (in brief): BA/BE including food effect, dissolution/IVIVC/BCS, and clinical PK assay summaries (LLOQ/ULOQ, A/P, stability, […]
From Bioanalysis to Biowaivers: Writing Module 2.7.1 That Travels From IND to NDA Read More »