CTD Series – Module 2.7.2: Building a Label-Ready Dose Rationale from IND to NDA
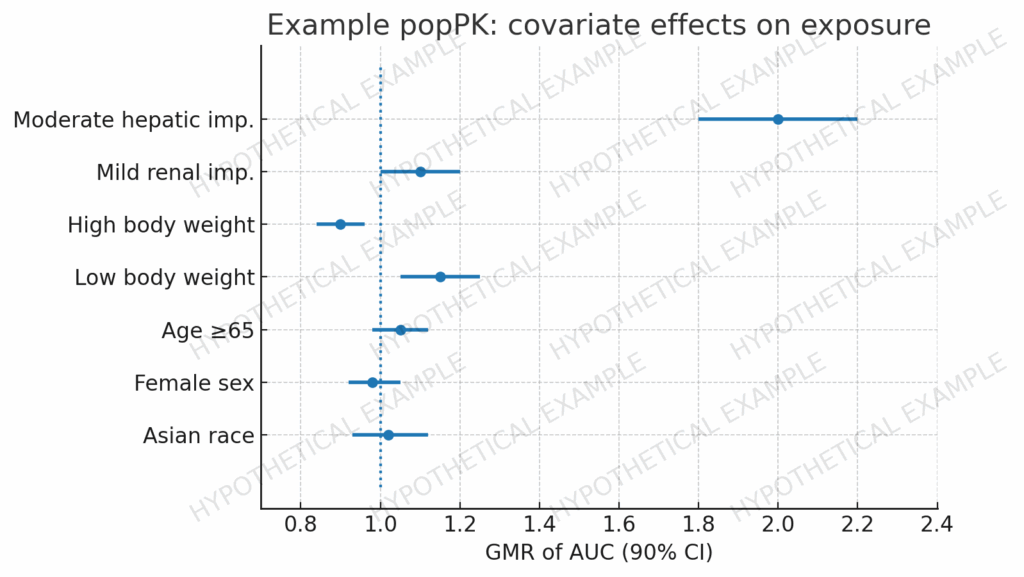
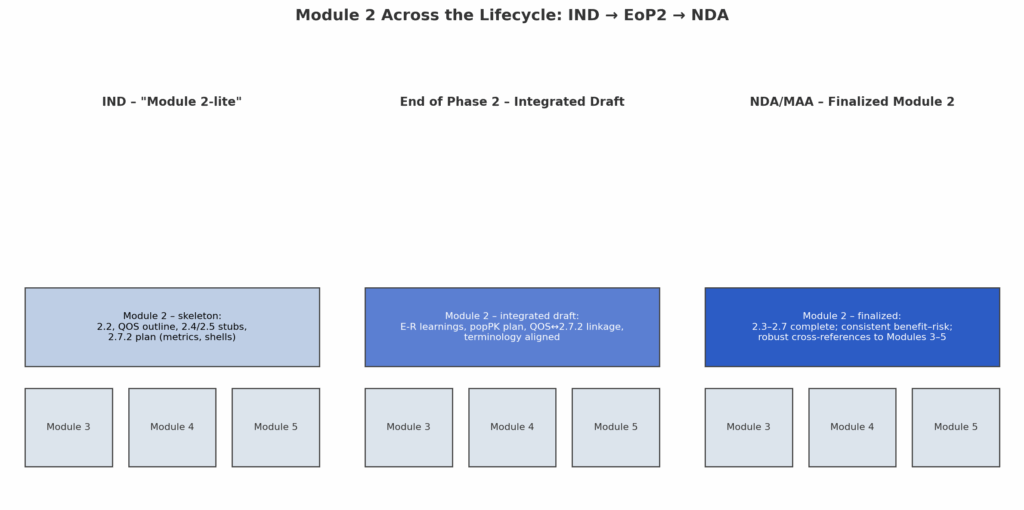
Dossier Deep Dive · Part 3 Executive Snapshot Why 2.7.2 matters: This is the concise, integrated clinical pharmacology narrative that underpins dose, regimen, and labeling—read alongside the Clinical Overview (2.5) and substantiated by Module 5. Start at IND: Draft a light “2.7.2” scaffold (exposure metrics, covariates, DDI/food-effect plan, modeling shells) and harden iteratively through EoP2. […]
CTD Series – Module 2.7.2: Building a Label-Ready Dose Rationale from IND to NDA Read More »