Many 2026 INDs are already being scoped. Budgets are being finalized, CROs are being shortlisted, and timelines are being compressed. In that environment, clinical pharmacology often becomes something to be delivered rather than something to be designed. Analyses are commissioned because the IND needs them, not because a decision requires them.
The risk here is subtle. Programs rarely fail because a model is missing. They struggle because decisions are made without a coherent exposure framework, and the gaps only become visible when regulators, internal governance, or downstream trials start asking uncomfortable questions. The biggest risk is not what is absent. It is what is disconnected.
Clinical Pharmacology Is Meant to Be the Decision Spine of the Program
At its core, clinical pharmacology exists to support decisions: What dose should enter humans? How should escalation proceed? What exposures define acceptable safety margins? When done well, clinical pharmacology connects preclinical evidence, early clinical data, and study design into a single, defensible narrative [1].
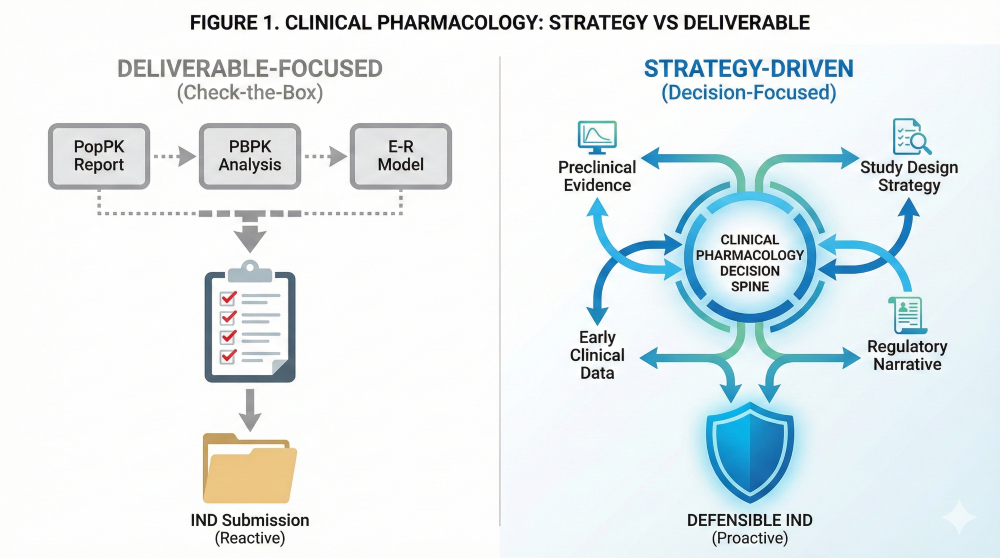 Figure 1. Clinical Pharmacology as Strategy vs. Deliverable framework.
Figure 1. Clinical Pharmacology as Strategy vs. Deliverable framework.
When treated as a deliverable, it becomes a collection of outputs that are technically correct but strategically thin. Figure 1 contrasts a deliverable-focused, “check-the-box” approach with a strategy-driven, decision-focused framework.
How Deliverable-Driven Thinking Creeps In
Most teams do not set out to fragment their programs; the drift happens gradually. Modeling is often commissioned late because “the IND needs it,” or different vendors generate parallel analyses with little alignment. This is often driven by the FDA’s Project Optimus initiative, which now mandates randomized dose-finding trials that many teams are not structurally prepared to execute [1].
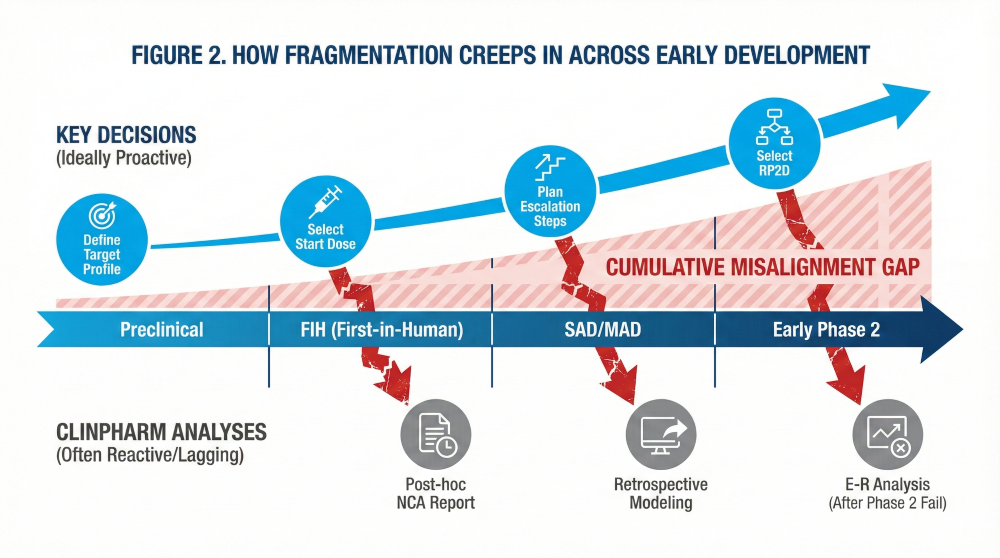 Figure 2. How Fragmentation Creeps In Across Early Development.
Figure 2. How Fragmentation Creeps In Across Early Development.
Figure 2 illustrates how analyses lag behind key decisions across preclinical, FIH, SAD/MAD, and early Phase 2, creating cumulative misalignment that regulators eventually flag during BLA/NDA reviews [2].
Early Choices That Are Hard to Fix Later
Some decisions appear small but quietly lock programs into constrained paths. Dose escalation schemes chosen before exposure targets are articulated or sparse sampling designs that limit exposure–response interpretability can become expensive roadblocks. Once these choices are embedded, “fixing it later” is often impossible.
 Figure 3. Early Decisions That Lock in Long-Term Consequences.
Figure 3. Early Decisions That Lock in Long-Term Consequences.
What Regulators Actually Notice
Regulators do not expect perfection, but they do notice coherence. They look for consistency across dose rationale, study design, and safety margins. They notice when Modules 2 and 5 tell slightly different stories [3].
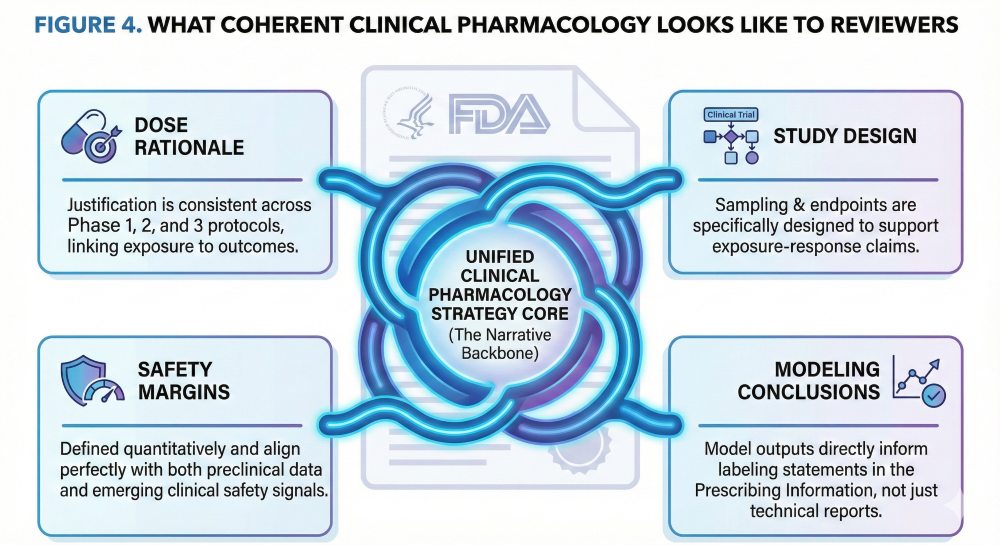 Figure 4. What Coherent Clinical Pharmacology Looks Like to Reviewers.
Figure 4. What Coherent Clinical Pharmacology Looks Like to Reviewers.
Signs Your Program Is Slipping into Reactive Mode
| Aspect | Deliverable-Focused | Strategy-Focused |
|---|---|---|
| Primary goal | Submission completeness | Decision quality |
| Timing | Late, reactive | Early, proactive |
| Role of modeling | Descriptive | Prescriptive |
| Narrative | Fragmented | Coherent |
| Signal | What It Often Indicates |
|---|---|
| Modeling starts after decisions are made | Strategy defined too late |
| Analyses cannot be reused | Lack of upfront alignment |
| Dose justification feels defensive | Exposure framework unclear |
Reframing Clinical Pharmacology as a Strategy
Successful programs define decisions before defining analyses. They use clinical pharmacology to reduce uncertainty, not just to decorate narratives. This approach often saves time by preventing late-stage rework, such as the dosage optimization issues seen in recent Oncology filings [3] [4].
Conclusion: The Quiet Risk Is Avoidable
Treating clinical pharmacology as a strategy rather than a deliverable does not require more analysis. It requires clearer thinking. Programs succeed when ClinPharm functions as the connective tissue across decisions, data, and regulatory communication.
How We Help
We work with early and mid-stage teams to structure clinical pharmacology as a decision-making framework. Our focus is on defining the right questions, aligning analyses across stages, and maintaining a coherent regulatory narrative through IND and beyond.
Optimize Your 2026 StrategyAbbreviations
- BLA: Biologics License Application
- CRO: Contract Research Organization
- FIH: First-in-Human
- IND: Investigational New Drug
- MAD: Multiple Ascending Dose
- NDA: New Drug Application
- PK/PD: Pharmacokinetics/Pharmacodynamics
- SAD: Single Ascending Dose
References
- FDA Draft Guidance (2023). Optimizing the Dosage of Human Prescription Drugs for Oncologic Diseases.
- Pfizer Portfolio Analysis (2025). Impact of MIDD on Cycle Times and Costs. CP&T.
- FDA ODAC Briefing Document (2023). Sotorasib Dosage Optimization Analysis.
- Yu, Y. et al. (2017). PBPK Modeling of Palbociclib to Support DDI Labeling. J. Clin. Pharmacol.
