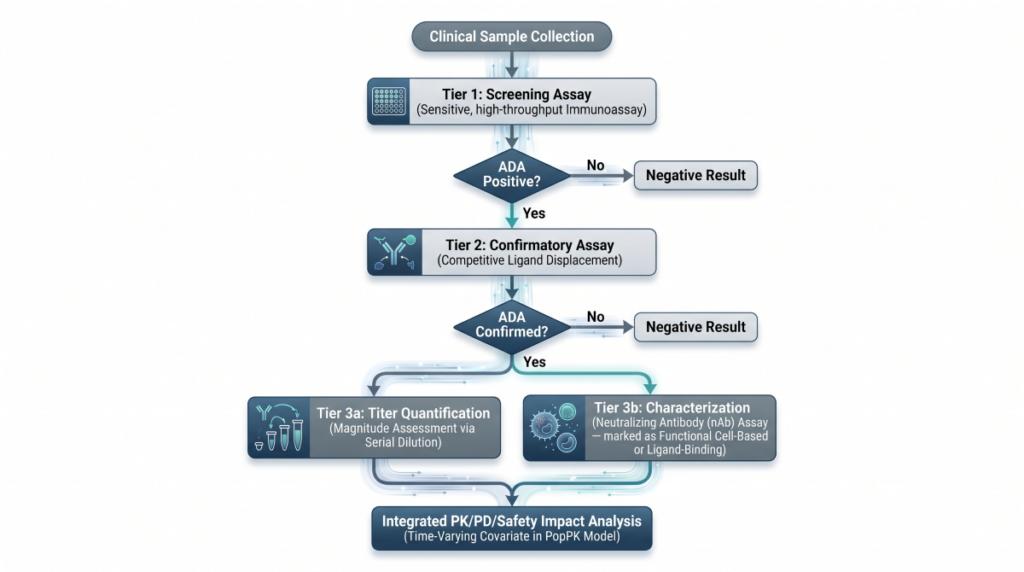
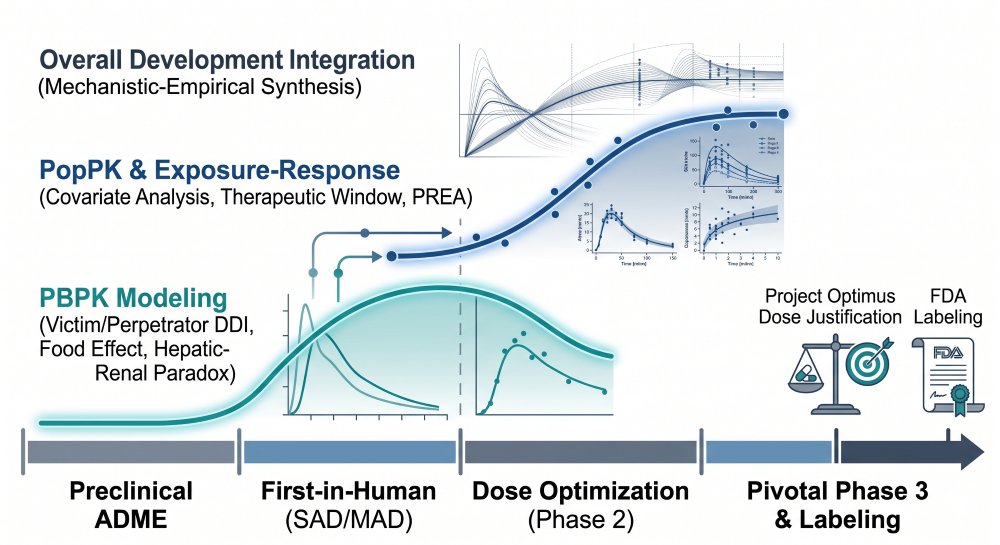
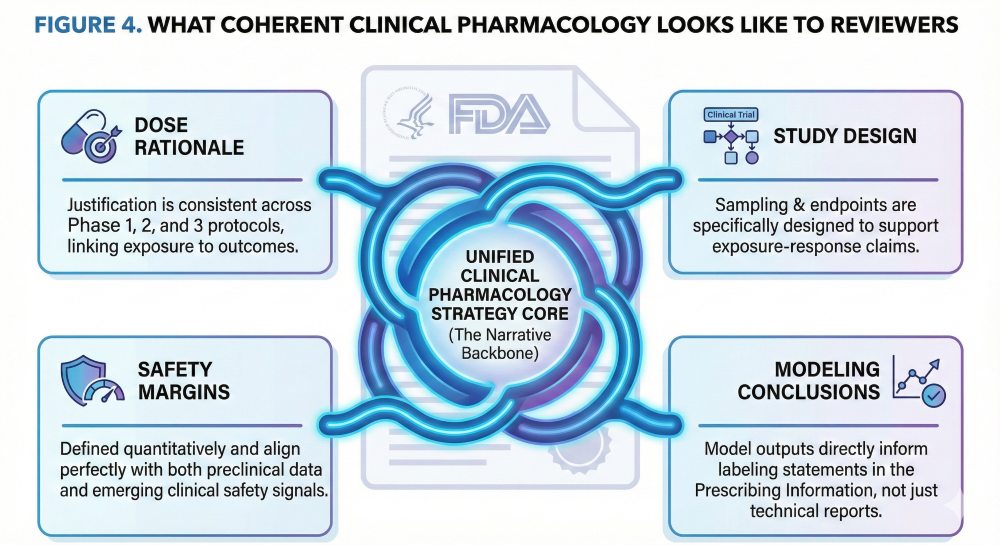
The Large Molecule Clinical Pharmacology Plan: Mechanistic Disposition of Monoclonal and Bispecific Antibodies
The Modality Matrix Series Portfolio This third report shifts focus from low-molecular-weight chemical entities to engineered macromolecular biologics. The clinical pharmacology plan (CPP) for large molecules requires multi-dimensional characterization of tissue convection, target-mediated saturation, spatial binding kinetics, and immunogenicity profiles to clear modern regulatory barriers. Article Sequence Thematic Focus Status Article 1 Overview of Modalities […]