Executive Summary: As of March 2026, pediatric drug development has moved beyond simple “trial size reduction” to a strategy of Mechanistic Justification. While the finalized ICH E11A (2025) guideline provides the high-level extrapolation tree, the February/March 2026 FDA Modernization Pack—specifically the Plausible Mechanism Framework and the new Bayesian Methodology Standards—has redefined the “totality of evidence.” In 2026, success in pediatric submissions, particularly in ultra-rare indications ($N < 10$), is predicated on using Physiologically Based Pharmacokinetics (PBPK) as the specific mathematical proof of biological causation.
1. The ICH E11A Continuum: The Foundation
The finalization of ICH E11A (effective January 2025) replaced rigid extrapolation categories with an Extrapolation Continuum. This framework recognizes that pediatric evidence is not a binary choice but a spectrum based on the “certainty of similarity.”
- Disease Similarity is Paramount: The initial step remains proving that the disease pathophysiology and therapeutic response are consistent between adults and children.
- The Role of Population PK (Pop-PK): Standard Pop-PK is utilized to bridge exposures. If disease similarity is accepted, Pop-PK proves that the pediatric dose achieves the same systemic exposure known to be effective in adults.
2. PBPK: The Mechanistic Anchor for Individualized Therapies (2026 Update)
While ICH E11A provided the continuum, the February 2026 FDA Draft Guidance: “Establishing a Plausible Mechanism Supporting Approval of Individualized Therapies” introduced a critical pivot for ultra-rare pediatric indications. For these therapies ($N < 10$), simple "exposure matching" via plasma levels is insufficient.
The FDA now requires a Mechanistic Anchor—a mathematical proof that the therapy is engaging the molecular root cause of the disease. PBPK modeling has emerged as the required engine for this framework for two primary reasons:
- • Site of Action Proof: PBPK predicts the local concentration at the specific cellular site of action (e.g., CNS tissue or hepatic intracellular space), proving the drug is present at the target in therapeutic concentrations, not just circulating in the blood.
- • Ontogeny Scaling: PBPK accounts for the maturation of specific transporters and enzymes (ontogeny). This ensures the “Plausible Mechanism” remains valid as the child grows, moving beyond simple weight-based allometric scaling.
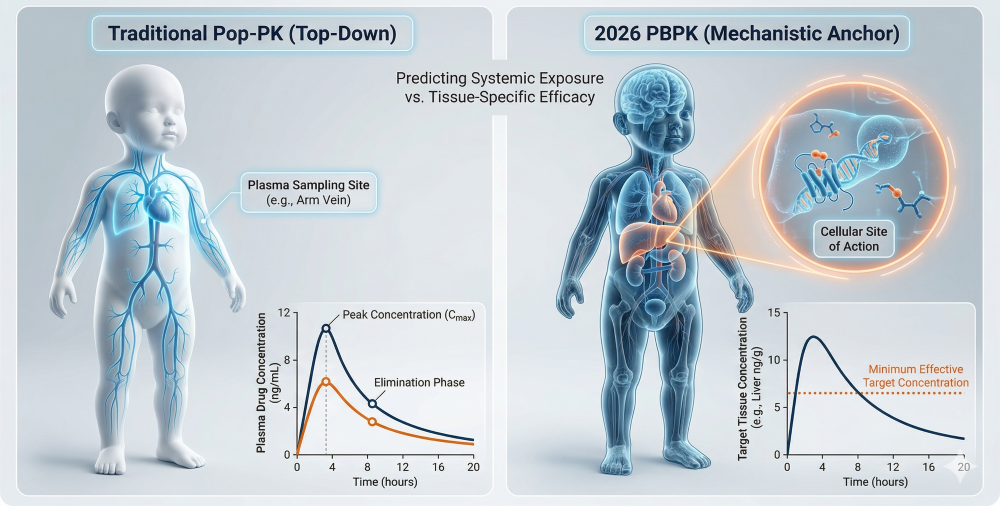
Figure 1: The PBPK Mechanistic Anchor — Proving Target Engagement at the Site of Action
3. Bayesian Methodology: The Evidence Engine (Jan 2026 Update)
Following the January 2026 FDA Bayesian Guidance, the mathematical “engine” for ICH E11A extrapolation has been standardized. This allows for formal Information Borrowing from adult data to supplement small pediatric cohorts.
- Prior Effective Sample Size (ESS): By calculating a clear ESS, a sponsor can determine exactly how many “virtual” adult patients equal one “real” pediatric patient in a statistical model.
- Statistical Power: In 2026, this settles the debate on trial power. A pediatric study of only 12 patients can be statistically “powered” to the equivalent of 100 patients using Bayesian priors derived from adult Phase 3 data.
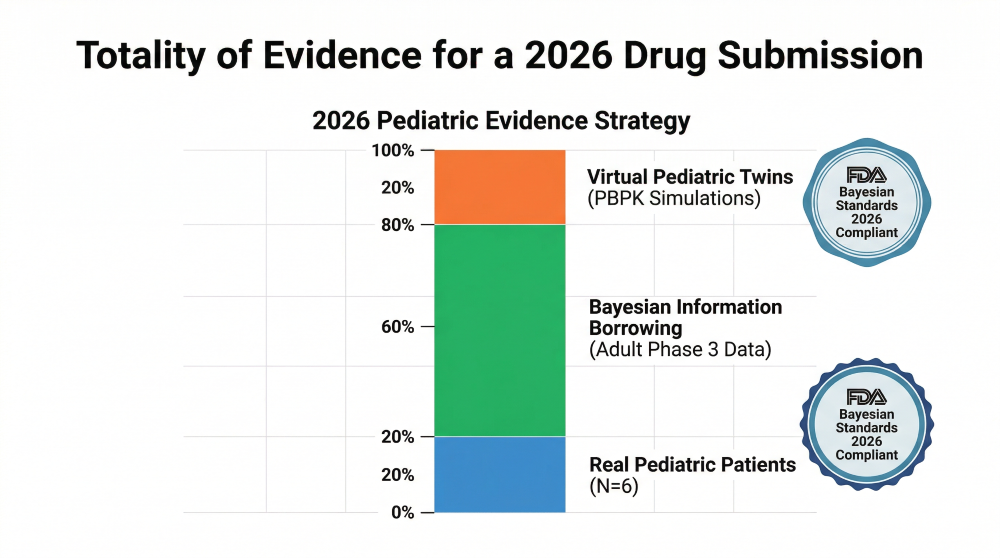
Figure 2: Powering the Trial — Bayesian Evidence Borrowing from Adult Priors and Virtual Twins
4. The 2026 Shift to “Mechanistic Safety”
While 2024 focused primarily on efficacy, 2026 is the year of Mechanistic Safety Extrapolation. Regulators now expect PBPK models to address Secondary Pharmacology. By utilizing ontogeny-informed models, sponsors must predict potential “off-target” effects on developing organ systems (like neurodevelopment or bone growth) that might not be visible in adult safety data. Safety is no longer just monitored; it is mechanistically predicted.
5. The 2026 Pediatric Decision Framework
Navigating these overlapping guidances requires a unified approach. The 2026 workflow integrates the ICH E11A “Extrapolation Tree” with the specific mechanistic and Bayesian requirements for individualized therapies.
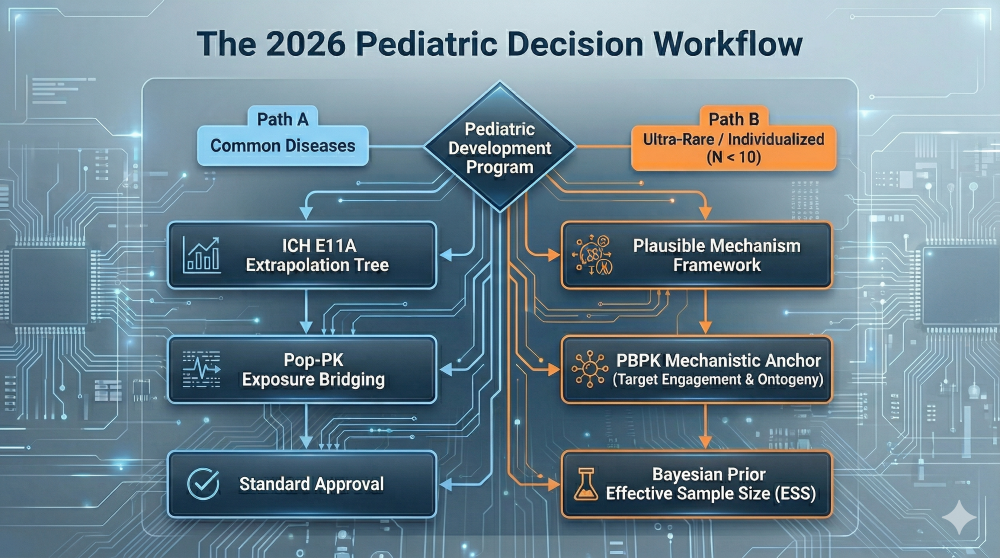
Figure 3: The 2026 Pediatric Decision Framework — Integrating ICH E11A and the Plausible Mechanism Framework
6. Summary: The 2026 Regulatory Landscape
| Guidance Feature | 2024 (ICH E11A) | 2026 Regulatory Modernization Pack |
|---|---|---|
| Primary Goal | Trial size reduction | Bridging via Biological Causation |
| Ultra-Rare Strategy | Case-by-case discussion | Plausible Mechanism Framework (PBPK-based) |
| Evidence Basis | PK Bridging (Pop-PK) | “White Box” PBPK Target Engagement |
| Statistical Engine | Frequentist / Basic PK | Bayesian / Prior Borrowing Standards |
Selected References
- ICH E11A Guideline (2024). Pediatric Extrapolation. Step 4.
- FDA Draft Guidance (Feb 2026). Establishing a Plausible Mechanism Supporting Approval of Individualized Therapies.
- FDA Guidance (Jan 2026). Bayesian Statistical Methods in Drug Development: Traceability and Prior Borrowing.
- Kuepfer, L. et al. (2025). Applied Concepts in PBPK: Transparency and the Open Systems Pharmacology Suite.
Expert FAQ: ICH E11A & 2026 Modernization
ICH E11A (2024) is the general framework. The 2026 pack (specifically the Plausible Mechanism Framework and Bayesian guidance) provides the “mathematical bridge” for the extreme end of that framework—allowing approval based on target engagement and mechanistic proof when RCTs are impossible ($N < 10$).
Individualized pediatric therapies often address genetic root causes at the cellular site of action. Pop-PK only measures blood levels. PBPK is the only tool that can simulate local tissue concentrations and target engagement (TMDD) at the specific molecular site where the disease originates.
Virtual twins are simulated pediatric patients built from real-world physiological data. In 2026, they are used to stress-test your PBPK dosing assumptions and augment small clinical cohorts when recruiting rare pediatric patients is logistically or ethically difficult.
Yes. Regulators expect PBPK modeling to specifically screen for potential “off-target” effects on developmental organ systems (like neurodevelopment), rather than relying solely on adult safety extrapolation.
Easier, but only if you have robust adult data. By standardizing “Prior Effective Sample Size” calculations, the 2026 guidance allows sponsors to calculate exactly how many “virtual” adult patients equal one “real” pediatric patient.
